Indexed In
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Access to Global Online Research in Agriculture (AGORA)
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- SWB online catalog
- Publons
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
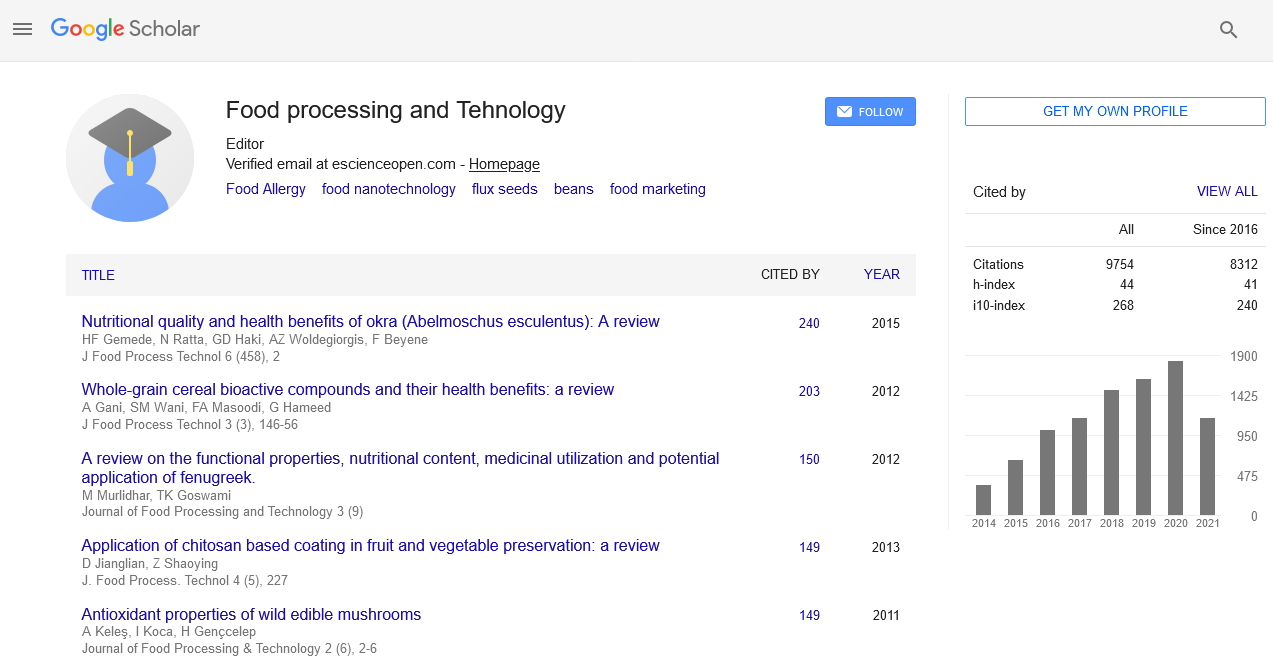
Validation of multi-residue screening method for antibiotics in livestock using liquid chromatography tandem mass spectrometry
3rd European Food Safety & Standards Conference
October 24-25, 2016 Valencia, Spain
Kim Min Kyoung, Kim Byung Joon, Wang So young and Kim Do Hun
Ministry of Food and Drug Safety, Korea
Posters & Accepted Abstracts: J Food Process Technol
Abstract:
A fast and simple method for residue analysis of the 90 antibiotics was validated for beef, pork, chicken muscle, milk and eggs. For the sample preparation, 2 g of livestock sample was extracted with acetonitrile, C18 and hexane was added to remove matrix and fat. After evaporation, sample was dissolved in methanol and DW containing 0.1% of formic acid. Liquid chromatography tandem mass spectrometry was used for instrumental analysis and 2.0 X150 mm, 3 μm columns was used. Mobile phases were composed by acetonitrile and DW with 0.1% formic acid. For the linearity, concentration ranged from 0.0002-1.2 ugg-1. Limits of detection were from 0.1-40 ngmL-1. On the recovery test, more than 87% of components meet the requirements of 50 ~ 120% recovery range and less than 35 CV. After the validation process, 130 representative samples are collected from five regions (Seoul, Daegu, Busan, north of Gyeonggi province and Gangwon province) and all the samples are analyzed based on validated method.
Biography :
Kim Min Kyoung has completed her PhD from Chonnam University, her major is Veterinary Medicine. She is the Scientific Officer in Seoul Regional Food & Drug Administration, Ministry of Food and Drug Safety.
Email: mg6153542@korea.kr