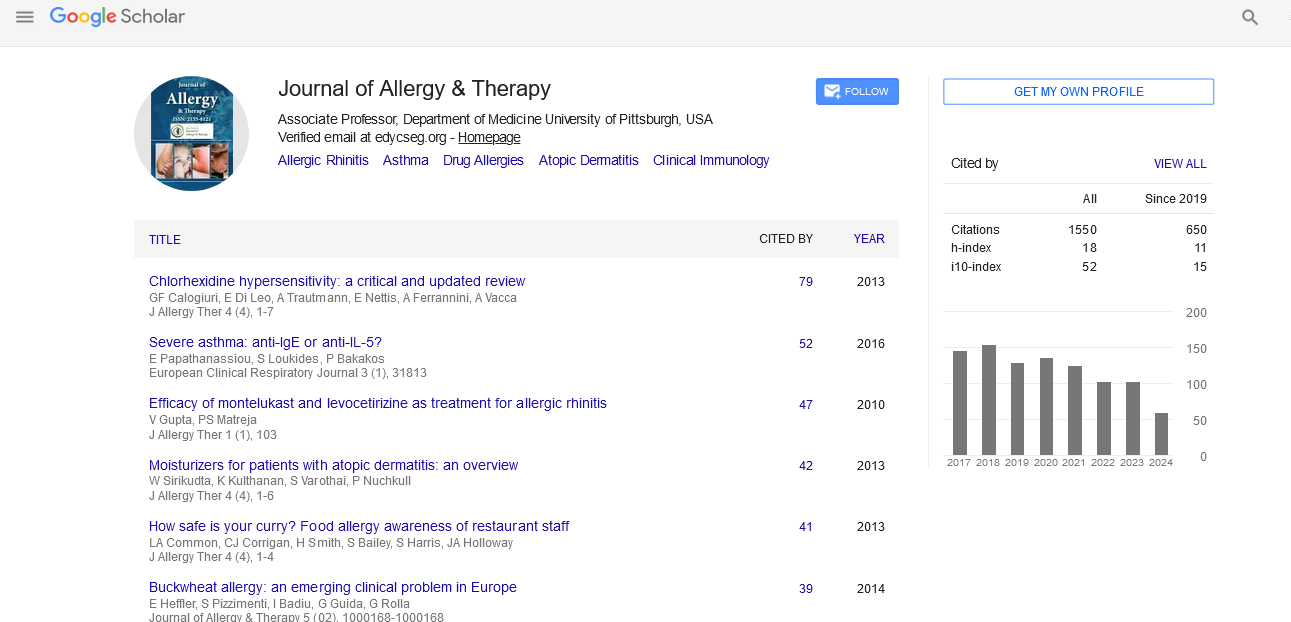
Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Short Communication - (2025) Volume 16, Issue 4
Therapeutic Strategies and Immune Responses in Allergen Immunotherapy
Elena Petrova*Received: 24-Nov-2025, Manuscript No. JAT-25-31333; Editor assigned: 26-Nov-2025, Pre QC No. JAT-25-31333 (PQ); Reviewed: 10-Dec-2025, QC No. JAT-25-31333; Revised: 17-Dec-2025, Manuscript No. JAT-25-31333 (R); Published: 24-Dec-2025, DOI: 10.35248/2155-6121.25.16.447
Description
Allergen Immunotherapy is a medical treatment designed to modify the immune system's response to allergens and provide long-term relief from allergic diseases. It is widely used for conditions such as allergic rhinitis, allergic conjunctivitis, asthma and insect venom allergy. The therapy works by gradually exposing patients to increasing doses of the specific allergen responsible for their symptoms, with the goal of inducing immune tolerance. Unlike conventional pharmacological treatments that primarily manage symptoms, allergen immunotherapy targets the underlying immunological mechanisms that drive allergic reactions. This approach has been shown to reduce symptom severity, decrease the need for medications and in some cases, prevent the progression of allergic diseases. The treatment can be administered via subcutaneous injections or sublingual tablets or drops, depending on the allergen and patient characteristics, with both methods demonstrating efficacy in clinical practice.The immunological basis of allergen immunotherapy involves a complex interplay between innate and adaptive immune responses. Repeated exposure to controlled doses of allergens leads to the modulation of T helper cell activity, specifically reducing the dominance of T helper type 2 cells that drive IgE mediated reactions. This shift promotes the activation of regulatory T cells and B cells, which produce Immunoglobulin G4 antibodies that can block the interaction between allergens and IgE. Consequently, mast cells and basophils are less likely to release histamine and other inflammatory mediators upon allergen exposure. Over time, these immunological changes result in reduced hypersensitivity and improved clinical tolerance. Research has also shown that allergen immunotherapy can lead to long-term modifications in immune memory, which may explain its sustained benefits even after the discontinuation of therapy [1-5].
Clinical indications for allergen immunotherapy are determined based on patient history, diagnostic testing and severity of symptoms. It is particularly recommended for patients with moderate to severe allergic rhinitis or asthma who do not respond adequately to conventional medications. In cases of venom allergy, immunotherapy is the treatment of choice due to its ability to prevent life-threatening anaphylactic reactions. Prior to initiating therapy, patients undergo a thorough evaluation, including skin prick testing and measurement of allergen-specific immunoglobulin E, to identify the causative allergens. The dosing schedule is carefully tailored to each patient, with an initial build-up phase followed by a maintenance phase that can last several years. Monitoring during therapy is essential to manage potential adverse reactions, which can range from mild local swelling to, in rare cases, systemic reactions requiring immediate intervention. Safety considerations are central to the administration of allergen immunotherapy. Subcutaneous therapy carries a risk of systemic allergic reactions, including anaphylaxis, which necessitates administration in a controlled medical setting with appropriate monitoring and emergency management. Sublingual immunotherapy has a more favorable safety profile, with most reactions being localized and mild, such as oral itching or swelling. Patient selection, adherence to dosing schedules and education regarding potential side effects are critical components of safe and effective treatment. Ongoing research aims to optimize formulations, dosing strategies and delivery methods to enhance efficacy while minimizing risks, including the exploration of adjuvants and novel allergen preparations that may improve immune tolerance induction.
The impact of allergen immunotherapy extends beyond individual patients, with significant implications for healthcare systems and public health. By reducing reliance on long-term pharmacotherapy, hospital visits and management of complications, immunotherapy can lower healthcare costs and improve quality of life. It also emphasizes a personalized approach to medicine, tailoring interventions to the specific allergens and immune profiles of each patient. Integration of immunotherapy with conventional treatments, lifestyle modifications and environmental control measures provides a comprehensive strategy for managing allergic diseases and achieving sustained clinical benefit [6-10].
Conclusion
In allergen immunotherapy represents a transformative approach in the management of allergic diseases, targeting the underlying immune mechanisms responsible for hypersensitivity reactions. By inducing immune tolerance and modulating the activity of key immune cells and antibodies, it offers long-term relief from symptoms and the potential to alter the course of allergic conditions. Both subcutaneous and sublingual administration methods are effective, with careful patient selection and monitoring ensuring safety. Beyond individual patient outcomes, allergen immunotherapy contributes to broader public health benefits by reducing disease burden and healthcare costs. Continued research and refinement of immunotherapy protocols hold promise for enhancing efficacy, expanding indications and improving patient experience, solidifying its role as a cornerstone in modern allergy treatment.
References
- Ring J, Gutermuth J. 100 years of hyposensitization: history of allergenâ?specific immunotherapy (ASIT). ALGY. 2011;66(6):713-724.
[CrossRef] [Google Scholar] [PubMed]
- Maggi E. T cell responses induced by allergen-specific immunotherapy. Clin Exp Immunol. 2010;161(1):10-18.
[CrossRef] [Google Scholar] [PubMed]
- Akdis M, Akdis CA. Mechanisms of allergen-specific immunotherapy: multiple suppressor factors at work in immune tolerance to allergens. J Allergy Clin Immunol. 2014;133(3):621-631.
[CrossRef] [Google Scholar] [PubMed]
- Pitsios C, Demoly P, Bilò MB, Gerth van Wijk R, Pfaar O, et al. Clinical contraindications to allergen immunotherapy: an EAACI position paper. ALGY. 2015;70(8):897-909.
[CrossRef] [Google Scholar] [PubMed]
- Huang HJ, Resch-Marat Y, Rodriguez-Dominguez A, Chen KW, Kiss R, et al. Underestimation of house dust mite–specific IgE with extract-based ImmunoCAPs compared with molecular ImmunoCAPs. J Allergy Clin Immunol. 2018;142(5):1656-1659.
[CrossRef] [Google Scholar] [PubMed]
- Kim EH, Patel C, Burks AW. Immunotherapy approaches for peanut allergy. Expert Rev Clin Immunol. 2020;16(2):167-174.
[CrossRef] [Google Scholar] [PubMed]
- Villalba M, Rodríguez R, Batanero E. The spectrum of olive pollen allergens. From structures to diagnosis and treatment. Methods. 2014;66(1):44-54.
[CrossRef] [Google Scholar] [PubMed]
- Gregory LG, Lloyd CM. Orchestrating house dust mite-associated allergy in the lung. Trends Immunol. 2011;32(9):402-411.
[CrossRef] [Google Scholar] [PubMed]
- Lourenco LO, Ribeiro AM, Lopes FD, Tibério ID, Tavares-de-Lima W. Different phenotypes in asthma: clinical findings and experimental animal models. Clin Rev Allergy Immunol. 2022;62(1):240-263.
[CrossRef] [Google Scholar] [PubMed]
- Kuruvilla ME, Lee FE, Lee GB. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clin Rev Allergy Immunol. 2019;56(2):219-233.
[CrossRef] [Google Scholar] [PubMed]
Citation: Petrova E (2025) Therapeutic Strategies and Immune Responses in Allergen Immunotherapy. J Allergy Ther.16:447.
Copyright: © 2025 Petrova E. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.