Indexed In
- Academic Journals Database
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Scimago
- Access to Global Online Research in Agriculture (AGORA)
- Electronic Journals Library
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- MIAR
- University Grants Commission
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2023) Volume 15, Issue 2
The Helpful Effect of Vitamin D and Coconut Oil in Modulating the Histological Disorders of the Splenic Tissue in Hyperglycemic Mice
Nabila I El-Desouki*, Mohamed L Salem, Dalia F Afifi, Nasef M and Faten M AbdallahReceived: 11-Mar-2023, Manuscript No. JMBT-23-20110; Editor assigned: 13-Mar-2023, Pre QC No. JMBT-23-20110 (PQ); Reviewed: 27-Mar-2023, QC No. JMBT-23-20110; Revised: 22-May-2023, Manuscript No. JMBT-23-20110 (R); Published: 29-May-2023, DOI: 10.35248/1948-5948.23.15.548
Abstract
The present study aims to study the effect of Vitamin D (Vit D) or/and Coconut Oil (Coc) on the splenic histological changes of diabetic adult mice induced by STZ. The mice were divided into 7 groups, and the experimental duration was 4 weeks. Group I: Control group without any treatments; group II and group III: Non-diabetic groups orally received vitamin D in a dose of 500 IU (6.25 ml)/kg bw/d or Coc in a dose of 7.5 ml/kg bw/d; group IV: Diabetic group
injected IP with a single dose of STZ (200 mg/kg bw); groups V, VI and VII: Administration of vitamin D or Coc or both together to diabetic group. The results recorded: Group II and group III no significant changes in the Blood Glucose (BG), insulin splenic weight levels. Group IV had a significant increase in BG, significant decrease in insulin and splenic weight values. Group V recorded a modest decrease in BG, and a modest rise in insulin andmarked splenic weight atrophy; while group VI or group VII registered a marked decrease in BG, a rise in insulin levels a marked splenic weight increment. Histologically, the splenic sections of control or non-diabetic mice received either vitamin D or Coc demonstrated normal structure of the solenocytes. Group IV showed great numbers of giant cells, disarrangement and interference of red and white pulps, and dilated congested blood vessels. Little improvement was seen in group V while group VI or group VII showed a marked improvement in the splenic tissues. In conclusion, diabetic mice received either Coc or co-dministered with vitamin D demonstrated strong anti-hyperglycemic effects to recovery the glucose and insulin rates to typical levels, and restored the splenic weight and histological architecture to normal status than those given vitamin D alone.
Keywords
Hyperglycemia; Spleen; Histology; Glucose; Insulin; Vitamin D; Coconut; Mice
Introduction
Hyperglycemia, the hallmark of the most prevalent and dangerous metabolic illness in humans known as Diabetes Mellitus (DM) disorders, is brought on by a variety of biochemical and physiological changes increased levels of free fatty acids, postprandial and/or fasting insulin, and pro-insulin, elevated intestinal glucose absorption, elevated hepatic glucose output, elevated peripheral glucose transport with increased glucose utilization are all connected [1]. Further, other symptoms such as increased insulin degradation and increased glucagon secretion enhanced catecholamines and insulin accelerated atherosclerosis development, and cardiovascular risk are also recorded [2].
Egypt had the eighth highest rate of diabetes in the world as of 2013. 15.6% of all adults in Egypt between the ages of 20 and 79 had type 2 diabetes in 2015 [3]. According to the world bank, this ratio was 16.7%. In 2017, there were 8.222.600 cases of diabetes in Egypt, and this number is expected to double by 2035 [4].
The spleen is the largest secondary lymphoid organ in the body and plays the main role in immune functions besides its roles in hematopoiesis and red blood cell clearance [5]. All vertebrates have the spleen. It recycles iron while discarding old red blood cells and keeping a blood reserve that might be useful in the case of hemorrhagic shock. It destroys hemoglobin that has been taken out of senescent red blood cells as a component of the mononuclear phagocyte system [6-10]. The heme portion of hemoglobin is converted into bilirubin, which is expelled in the liver, while the globin portion is reduced to its native amino acids [11]. The spleen produces antibodies in its white pulp and expels pathogens and antibody-coated blood cells through lymphatic and blood circulation. Because of how much more likely it is to get certain infections when it is absent, it might be compared to a large lymph node. According to a study on mice's spleen in 2009, more than half of the body's monocytes are found in the spleen's crimson pulp. When these monocytes move to injured tissue, such as the heart after a myocardial infarction, they develop into dendritic cells and macrophages that help the body repair.
The term "vitamin D" refers to a class of steroid hormones that are fat-soluble and boost calcium, magnesium, phosphate, and zinc absorption in the intestine. Different types of vitamin D exist (vitamers). There are two main types of vitamin D: Vitamin D2 (ergo-calciferol) and vitamin D3 (cholecalciferol). Without a subscript, vitamin D refers to calciferol, which is the chemical name for either vitamin D2 or D3 or both. The biological inactivation of vitamin D from the diet or cutaneous production from sunshine involves an enzymatic conversion (hydroxylation) throughout the kidney and liver. The body produces between 80% and 90% of the vitamin D it needs through skin synthesis, with the remainder coming from diet [12].
Vitamin D is required, according to some research, for healthy insulin production. Vitamin D's effects on insulin secretion and sensitivity may allow it to operate as a functional agent in the regulation of glucose tolerance [13]. Vitamin D insufficiency inhibits pancreatic insulin production, and this deficiency is linked to glucose intolerance and DMT2. Vitamin D supplementation in can be considered as a therapeutic agent to DMT2; it can improve the oxidation stress in diabetic patients and stimulates the insulin release.
Because it contains certain lipids that assist the body's natural hormone production and because it contains lauric acid, which is crucial for immunological support, coconut oil functions as hormonal support, capric acid and caprylic acid are advantageous for immune support since they have antifungal, antibacterial, and antiviral effects. Coconut oil is great for digestion since it contains a lot of healthy lipids. The best source of lauric acid is coconut oil [14]. Its antibacterial qualities can aid in the battle against candida infection and stomach discomfort. The antioxidant in coconut oil may reduce insulin resistance and harm to pancreatic beta cells in diabetics by scavenging Reactive Oxygen Species (ROS).
Some studies showed that coconut oil can help improve insulin levels when consumed regularly and increase absorption of calcium and magnesium. The superior effect of coconut oil over calcium is probably because it contains high amount of saturated fats in the form of medium chain triglycerides that are important for calcium absorption from the intestine [15]. Coconut oil supplementation in a diabetic patient can control the glycemic level.
The aim of the current work is to study the beneficial role of vitamin D or/and coconut oil on the blood glucose and insulin levels as well as in the weight and histopathology of the spleen of STZ diabetic adult male albino mice.
Materials and Methods
Animals
Vacsera in Cairo provided 70 mature male albino mice (Mus musculus), each weighing 25 ± 2 g and aged 6-8 weeks. For one week, the animals were acclimated in plastic cages (10 per cage) at the same temperature and with the same natural dark-light cycle. The animals had unrestricted access to food and water during the experiment. The institutional animal ethics committee of Tanta university approved all care and techniques used for the current experiment, and they also complied with guidelines for the humane handling and use of laboratory animals [16].
Induction of Diabetes Mellitus (DM)
STZ was acquired from "sigma chemicals co., St. Louis, Mo., USA" and dissolved in saline solution at a dose of 200 mg/ kg of body weight in order to induce DM in mice by Intraperitoneal (IP) injection.
Treatment
Vitamin D was obtained from "Egyptian group for pharmaceutical industries co., Egypt." and administered orally. Coconut oil was received from local pharmacy (Al-Badawia company for herbal and oil extraction, Mansoura, Egypt) and administered orally, by a gastric tube.
Experimental design
After acclimatization, the mice were divided into 7 equal groups (10 mice for each); all were kept under the same conditions and received the same diet. Group I: Normal control mice group without any treatments. Group II: Non-diabetic group received vitamin D orally in a dose of 500 international units (6.25 ml)/kg/bw/d for 4 weeks. Group III: 7.5 ml/kg bw of coconut oil was administered daily to the non-diabetic mouse group for 4 weeks. Group IV: The diabetic group received a single injection of STZ IP to cause diabetes as previously discussed. Group V: For four weeks, diabetic mice were given 500 international units (6.25 ml/kg body weight) of vitamin D orally. Group VI: Diabetic mice given orally coconut oil in a dose of 7.5 ml/kg/bw/d for 4 weeks. Group VII: Diabetic mice administered orally with both vitamin D and coconut oil at the same previous doses daily for 4 weeks.
Sample collection and sera separation
The animals were fasted for 14 hours at the conclusion of each session of the experiment, and were anaesthetized with diethyl ether, and then sacrificed [17]. All groups that were being researched had their blood drawn. Before centrifuging the blood for 20 minutes at 1000 rpm, the blood was allowed to clot for 30 minutes at room temperature. Serum samples were then taken and stored at 20°C until they were needed to assay insulin. The spleen specimens were removed and weighted then processed for histological study under light microscopy [18].
Histological study
Splenic specimens were fixed in 10% neutral buffered formalin for 24 hours, then they were processed in ethyl alcohol in progressive grades, cleared in xylene, embedded in paraffin wax, sectioned at a thickness of 5 μm, then stained with H and E to demonstrate the histological changes in accordance to Bancroft and Gamble.
Calculation of the results
For each pair of duplicate standards, control samples, and the average optical density of the zero standards, the mean absorbance was calculated. On a log-log graph paper, the standard curve was drawn with the common concentration on the x-axis and the absorbance on the y-axis. The best-fit straight line through the reference points was created considering Bourne [19].
Statistical analysis
The IBM SPSS® software package, version 16.0, USA, was used to enter data into the computer and analyse it. S.E. was used to the data analysis together with percentages and numbers. When the data were evenly distributed, the F-test (ANOVA) and post hoc test were employed to compare the seven research groups (LSD), according to significance was achieved at p 0.05.
Results
Effect of vitamin D and coconut oil on blood glucose levels
The induction of diabetes in adult mice by STZ caused highly significant increase in blood glucose level (group IV) as compared to the normal control mice (group I) (**p ≤ 0.001); a significant increase in blood glucose values of diabetic mice administered with vitamin D alone (group V) (*p ≤ 0.05) and non-significant increase in mice blood glucose levels of diabetic mice given coconut oil only (group VI) or co-administered with vitamin D and coconut oil together (group VII) as compared to the normal control mice (p>0.05), (Table 1 and Figure 1).
| Groups | Mean ± SE (mg/Di) |
|---|---|
| Group I (Normal control) | 83.9 ± 6.14 |
| Group II (Non-diabetic+Vit. D) | 79.4 ± 6.25 |
| Group III (Non-diabetic+CO) | 72.9 ± 7.03 |
| Group IV (DM) | 267.27 ± 35.87** |
| Group V (DM+Vit. D) | 202.2 ± 13.74* |
| Group VI (DM+CO) | 112.6 ± 9.68 |
| Group VII (DM+Vit. D+CO) | 87.1 ± 4.88 |
| Statistical significance was measured at p>0.05; *p ≤ 0.05;**p ≤ 0.005 | |
Table 1: Effect of vitamin D or/and coconut oil on blood glucose levels.
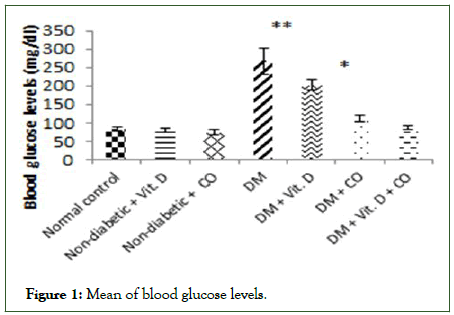
Figure 1: Mean of blood glucose levels.
Effect of vitamin D and coconut oil on insulin levels
Diabetic mice recorded a significant decrease in blood serum insulin (group IV) as compared to the normal control mice (group I) (*p ≤ 0.05), and non-significant decrease in blood serum insulin of the diabetic groups given vitamin D or/and coconut oil (groups V, VI and VII) as compared to the normal control mice (group I) (p>0.05), (Table 2 and Figure 2).
| Groups | Mean ± SE (ng/ml) |
|---|---|
| Group I (Normal control) | 1.27 ± 0.11 |
| Group II (Non-diabetic+Vit. D) | 1.41 ± 0.09 |
| Group III (Non-diabetic+CO) | 1.32 ± 0.11 |
| Group IV (DM) | 0.52 ± 0.14* |
| Group V (DM+Vit. D) | 0.72 ± 0.21 |
| Group VI (DM+CO) | 0.92 ± 0.22 |
| Group VII (DM+Vit. D+CO) | 1.17 ± 0.13 |
| Statistical significance was measured at p>0.05; *p ≤ 0.05 | |
Table 2: Effect of vitamin D or/and coconut oil on insulin levels.
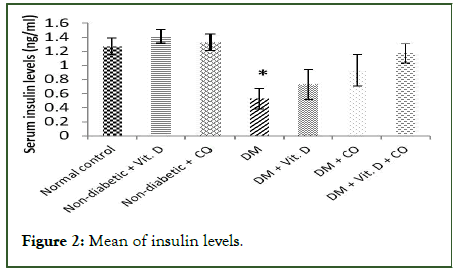
Figure 2: Mean of insulin levels.
Effect of vitamin D or/and coconut oil on spleen weight
The induction of diabetes in adult mice by STZ caused highly significant decrease in spleen weight values (Group IV) as compared to the normal control mice (group I) (**p ≤ 0.001); a significant decrease in spleen weight values of diabetic mice administered with vitamin D alone (group V) (*p ≤ 0.05) and non-significant decrease in spleen weight values of diabetic mice given coconut oil only (group VI) or given vitamin D and coconut oil together (group VII) as compared to the normal control mice (p>0.05), (Table 3 and Figure 3).
| Groups | Mean ± SE (mg) |
|---|---|
| Group I (Normal control) | 166.3 ± 3.74 |
| Group II (Non-diabetic+Vit. D) | 166.3 ± 2.65 |
| Group III (Non-diabetic+ CO) | 165.92 ± 3.27 |
| Group IV (DM) | 85.25 ± 5.41** |
| Group V (DM+Vit. D) | 138 ± 7.69* |
| Group VI (DM+CO) | 148.12 ± 5.57 |
| Group VII (DM+Vit. D+CO) | 148.75 ± 6.41 |
| Statistical significance was measured at p>0.05; *p ≤ 0.05;**p ≤ 0.001 | |
Table 3: Effect of vitamin D or/and coconut oil on spleen weight levels.
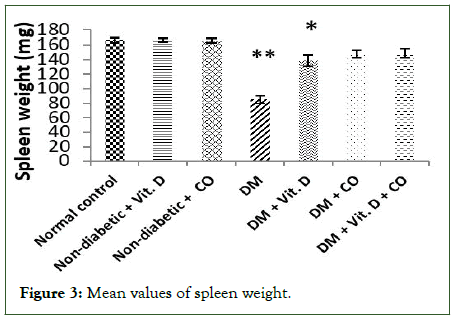
Figure 3: Mean values of spleen weight.
Histological observations
Normal control mice group (group I): The histological structure of spleen sections of a normal control mouse stained with H and E observed the normal structure of splenocytes with normal appearance of periarteriolar lymphoid sheaths of white pulp, red pulp and marginal zone between the red pulp and white-pulp [20]. Entire spleen is surrounded by the capsule which sends out trabeculae into the splenic parenchyma (Figure 4).
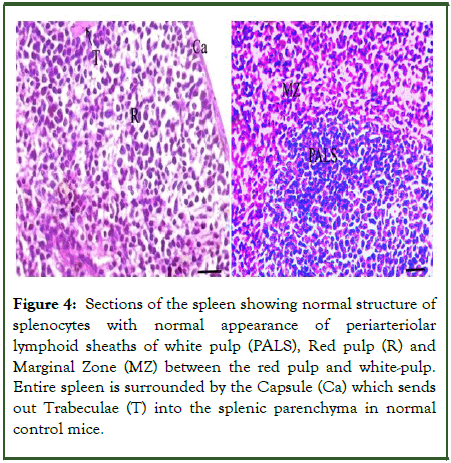
Figure 4: Sections of the spleen showing normal structure of splenocytes with normal appearance of periarteriolar lymphoid sheaths of white pulp (PALS), Red pulp (R) and Marginal Zone (MZ) between the red pulp and white-pulp. Entire spleen is surrounded by the Capsule (Ca) which sends out Trabeculae (T) into the splenic parenchyma in normal control mice.
Non-diabetic mice taken either vitamin D or coconut oil (groups II and III): The histological structures of the spleen sections of non-diabetic mice received vitamin D in a dose of 6.25 ml/kg b.w/d or coconut oil in a dose of 7.5 ml/kg b.w/d, respectively for 4 weeks illustrated similar normal structure as seen in normal control ones (Figures 5 and 6).
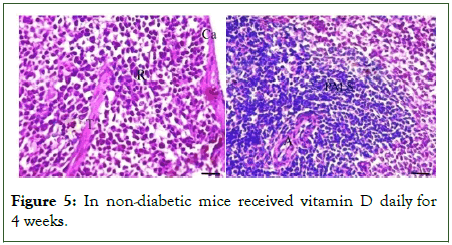
Figure 5: In non-diabetic mice received vitamin D daily for 4 weeks.
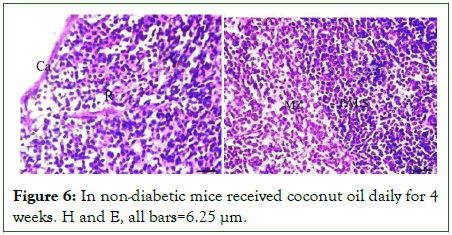
Figure 6: In non-diabetic mice received coconut oil daily for 4 weeks. H and E, all bars=6.25 μm.
STZ diabetic mice group (group IV): The diabetic mice group injected with a single dose of STZ (200 mg/kg/b.w) showed great numbers of giant cells, disarrangement and interference of red and white pulps between them. The dilated and congested blood vessels as well as thin capsule and thick branched trabeculae were also noticed (Figure 7).
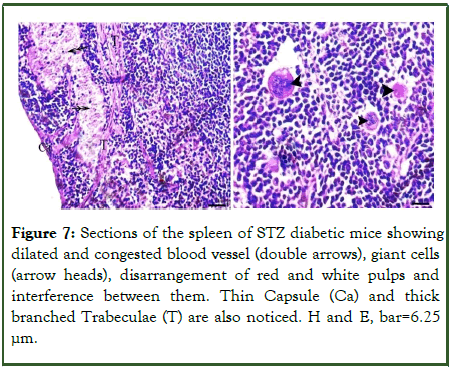
Figure 7: Sections of the spleen of STZ diabetic mice showing dilated and congested blood vessel (double arrows), giant cells (arrow heads), disarrangement of red and white pulps and interference between them. Thin Capsule (Ca) and thick branched Trabeculae (T) are also noticed. H and E, bar=6.25 µm.
The diabetic mice group given vitamin D (group V): The diabetic mice administered with vitamin D alone in a dose of 6.25 ml/kg b.w daily for 4 weeks demonstrated giant cells and interference between red and periarteriolar lymphoid sheaths of white pulps. Either normal thin capsule or trabeculae were seen (Figure 8).
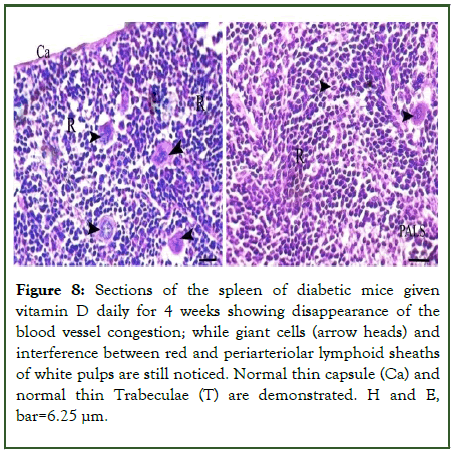
Figure 8: Sections of the spleen of diabetic mice given vitamin D daily for 4 weeks showing disappearance of the blood vessel congestion; while giant cells (arrow heads) and interference between red and periarteriolar lymphoid sheaths of white pulps are still noticed. Normal thin capsule (Ca) and normal thin Trabeculae (T) are demonstrated. H and E, bar=6.25 μm.
The diabetic mice groups given coconut oil alone or coadministered with vitamin D (groups VI and VII): The diabetic mice given coconut oil alone or co-administered with vitamin D daily for 4 weeks showed a marked recovery and improvement of red pulps, marginal zone and white pulps, and restored approximately their normal histological structure. Normal thin capsule and thin trabeculae were too illustrated (Figures 9 and 10).
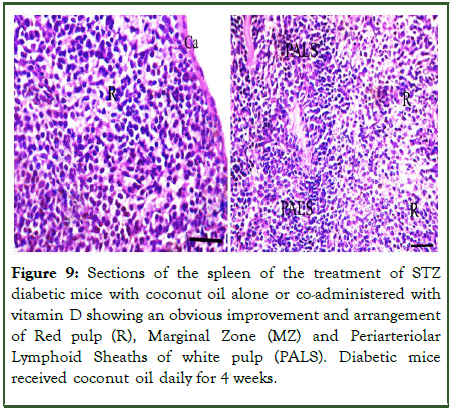
Figure 9: Sections of the spleen of the treatment of STZ diabetic mice with coconut oil alone or co-administered with vitamin D showing an obvious improvement and arrangement of Red pulp (R), Marginal Zone (MZ) and Periarteriolar Lymphoid Sheaths of white pulp (PALS). Diabetic mice received coconut oil daily for 4 weeks.
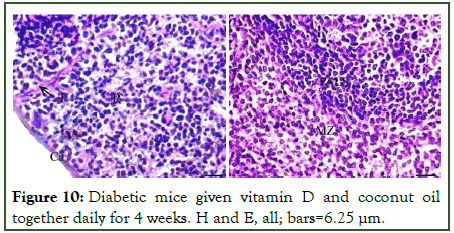
Figure 10: Diabetic mice given vitamin D and coconut oil together daily for 4 weeks. H and E, all; bars=6.25 μm.
Discussion
Diabetes Mellitus (DM) and its complications constitute a severe public health issue facing modern societies. A complete or partial lack of insulin secretion and/or activity causes problems in the metabolism of carbohydrates, proteins, and lipids. DM later leads to micro and macro-vascular complications and becomes a major cause of death.
The insulin producing beta cells of the pancreas in mammals are toxic to the glucosamine-nitrosourea compound Streptozotocin (STZ). By generating DNA damage, it is utilised in medical research to provide an animal model for hyperglycemia and in medicine to treat specific islets of langerhans malignancies.
In the results shown here, there were no appreciable differences in blood glucose and insulin levels between the non-diabetic adult mice groups. The STZ diabetic mice group had blood glucose levels that were substantially higher than those of the healthy control mice and insulin levels that were significantly lower. Compared to the group of diabetics, diabetic mice given coconut oil had a substantial decrease in blood glucose levels and a considerable increase in insulin, whereas diabetic mice given vitamin D demonstrated just a modest decrease in blood glucose levels and modest increases in insulin. The metabolic abnormalities caused by inadequate insulin secretion in type 2 diabetes range from hyperglycemia, which results from inadequate insulin-stimulated glucose uptake and increased hepatic glucose production, to dyslipidemia, which includes poor fatty acid, triglyceride, and lipoprotein homeostasis.
DM is a result of insulin resistance and vitamin D deficiency. The balance of glucose appears to be impacted by vitamin D. By reducing stress in the pancreatic cells and inhibiting the inflammatory responses brought on by cytokines, vitamin D prevents the apoptosis of pancreatic cells. Along with these molecular discoveries, there is a chance that vitamin D may play a part in the prevention of the onset of insulin resistance. Vitamin D ameliorated the harmful impact of diabetes mellitus, probably by increasing insulin secretion and sensitivity, ameliorating β-cell function, and decreasing cortisol levels, reduces the number of proinfammatory cytokines (e.g. IL-6), and increases the activity of the antioxidant system such as "GSH, SOD, TAC, and catalase" while reducing lipid peroxidation enzymes (e.g. MDA).
Coconut oil is a natural antioxidant that contains cotrienols, capric acid, caproic acid, and lauric acid. These compounds act as scavengers of potentially harmful oxygen free radicals, which are essential for the emergence of diabetes, ageing, atherosclerosis, and cancer. Coconut oil's lauric acid possesses characteristics that are insulin-tropic. Antioxidants may be extremely important for diabetes patients in enhancing insulin responsiveness to the loaded glucose and lowering insulin resistance.
The gradual decrease in blood glucose in STZ diabetic mice after being treated with coconut oil daily for 3 weeks has been recorded. Moreover, Iranloyel, et al., have observed that virgin coconut oil reduces hyperglycemia and enhances glucose tolerance, likely due to its antioxidant impact, which also enhances insulin production.
In the present study, the diabetic mice group recorded a marked atrophy in the weight of their spleen tissues but after taken vitamin D or coconut oil or both together daily for 4 weeks; the increment in the weight of mice spleen tissues was recorded and returned approximately to normal weight. Moreover, The present study's histology findings showed that normal control, non-diabetic mice given vitamin D or coconut oil had normal appearing red and white pulps with normal nuclei and a marginal zone separating the red pulp and white pulp when their spleen sections were stained with H and E. Entire spleen is surrounded by the capsule which sends out trabeculae into the splenic parenchyma. The diabetic mice group demonstrated a great numbers of giant cells, dilated and congested blood vessels, disarrangement of red and white pulps and interference between them. Thick branched trabeculae were also noticed. The diabetic mice that given vitamin D or/and coconut oil daily for 4 weeks noticed a marked improvement and recovery of approximately normal red pulps, marginal zone and white pulps.
Similar results were recorded in diabetic rats by Ebaid, et al. Hashish and Kamal illustrated the atrophy of diabetic splenic mice and diffusion of white pulp. Also, with dilated blood arteries, mature lymphocytes in the periphery of the spleen were too significantly decreased. In diabetic guinea pigs, similarly, Kumar, et al., found that the red pulp and white pulp frequently vacuolated with nuclei degeneration. The white pulp of the spleen resembles a lymph node structurally. It has T-cell and Bcell zones and enables the production of antigen specific immune responses that shield the body from bacterial, viral, and fungal infections that spread through blood. Immune responses that are harmful to the host can be controlled at the spleen.
Diabetes lowers immune response capabilities, including immune cell function reduction and immune organ atrophy. Additionally, diabetes and oxidative stress play a pivotal role in the development of diabetes complications and increase the cell death receptor Fas which caused splenocytes to die in rats via the Fas/FasL (programmed cell death receptors) pathway, suggesting a potential mechanism for the immunotoxicity of hyperglycemia.
The components of inflammation appear to be significantly impacted by vitamin D and its metabolites, insulin synthesis, secretion and action, and all of these may have an influence on the pathogenesis of DMT2. Through its immunomodulatory and anti-inflammatory properties, vitamin D appears to play a part in the pathogenesis of diabetes. By lowering insulin resistance, raising insulin production, and reducing autoimmune inflammation, vitamin D lowers the risk of developing diabetes types 1 and 2. Therefore, it is feasible that early vitamin D supplementation could provide protection against the onset of DM.
Virgin coconut oil contains the beneficial component lauric acid. Monolaurin, which has been demonstrated to influence immune cell proliferation and have antibacterial activity, is derived from lauric acid. Numerous more immune system functions are also involved in inflammation. For instance, during acute and chronic inflammations, in response to infections or other antigenic chemicals, immune cells become active. According to recent studies, inflammation contributes significantly to a number of human disorders that are not primarily immune system issues. These include Alzheimer's disease and other neurological diseases, ischemic heart disease, cancer, and atherosclerosis.
Conclusion
In conclusion, diabetic mice given either coconut oil alone or co-administered with vitamin D demonstrated strong antihyperglycemic effects to recovery the glucose and insulin rates to normal values, and restored the histological architecture of the spleen to approximately normal form than those given vitamin D alone.
Source of Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not for profit sectors.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Ethical Approval
Ethical approval was obtained from the public creature care and utilize council's convention and the rules for the consideration and utilization of exploratory creatures were followed all through the whole system.
Competing Interests
The authors have no conflict of interest with any other institutions.
Funding Source
The authors have no funding grants from any funding sources for this research work.
Author Contribution
The authors confirm that all persons designated as authors qualify for authorship and have verified the article for plagiarism. If plagiarism is detected, all authors will be held equally responsible and will bear the resulting sanctions imposed by the journal thereafter, M.L. conceived and designed the study, conducted research, F.A. provided research materials, and collected and organized data, D.F. analysed and interpreted data, wrote the initial and final drafts of the article, N.M. provided logistic support. N.I. critically reviewed and approved the final draft, and is responsible for the content and similarity index of the manuscript. And accountable for all aspects of the work.
References
- Abu-Taweel G, Ajarem JS, Hossam E, Rady AM. Behavioral changes induced by prenatal acute endotoxemia in mice offspring. Pakistan J Zool. 2013;45(2):359-369.
- Al-Malki AL, El Rabey HA. The antidiabetic effect of low doses of Moringa oleifera Lam. seeds on streptozotocin induced diabetes and diabetic nephropathy in male rats. Biomed Res Int. 2015;2015:381040.
[Crossref] [Google Scholar] [Pubmed]
- Al-Shoumer KA, Al-Essa TM. Is there a relationship between vitamin D with insulin resistance and diabetes mellitus? World J Diabetes. 2015;6(8):1057-1064.
[Crossref] [Google Scholar] [Pubmed]
- Baxter JD, Webb P. Thyroid hormone mimetics: Potential applications in atherosclerosis, obesity and type 2 diabetes. Nat Rev Drug Discov. 2009;8(4):308-320.
[Crossref] [Google Scholar] [Pubmed]
- Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev. 2020;16(5):442-449.
[Crossref] [Google Scholar] [PubMed]
- Berger MM, Shenkin A, Schweinlin A, Amrein K, Augsburger M, Biesalski HK, et al. ESPEN micronutrient guideline. Clin Nutr. 2022;41(6):1357-1424.
[Crossref] [Google Scholar] [Pubmed]
- Bergsson G, Steingrimsson O, Thormar H. Bactericidal effects of fatty acids and monoglycerides on Helicobacter pylori. Int J Antimicrob Agents. 2002;20(4):258-262.
[Crossref] [Google Scholar] [Pubmed]
- Boateng L, Ansong R, Owusu WB, Steiner-Asiedu M. Coconut oil and palm oil's role in nutrition, health and national development: A review. Ghana Med J. 2016;50(3):189-196.
[Google Scholar] [PubMed]
- Bronte V, Pittet MJ. The spleen in local and systemic regulation of immunity. Immunity. 2013;39(5):806-818.
[Crossref] [Google Scholar] [Pubmed]
- Deeds MC, Anderson JM, Armstrong AS, Gastineau DA, Hiddinga HJ, Jahangir A, et al. Single dose streptozotocin induced diabetes: Considerations for study design in islet transplantation models. Lab Anim. 2011;45(3):131-140.
[Crossref] [Google Scholar] [Pubmed]
- de Fronzo RA. Pathogenesis of type 2 diabetes mellitus. Med Clin North Am. 2004;88(4):787-835.
[Crossref] [Google Scholar] [Pubmed]
- Ebaid H. Promotion of immune and glycaemic functions in streptozotocin-induced diabetic rats treated with un-denatured camel milk whey proteins. Nutr Metab (Lond). 2014;11:31.
[Crossref] [Google Scholar] [Pubmed]
- Fatima El, Zahra MF, Sadek KM, Khafaga AF, Al Senosy AW, Ghoniem HA, Fayez S, et al. Vitamin D regulates insulin and ameliorates apoptosis and oxidative stress in pancreatic tissues of rats with streptozotocin induced diabetes. Environ Sci Pollut Res Int. 2022;29(60):90219-90229.
[Crossref] [Google Scholar] [PubMed]
- Girotti AW, Thomas JP. Damaging effects of oxygen radicals on resealed erythrocyte ghosts. J Biol Chem. 1984;259(3):1744-1752.
[Crossref] [Google Scholar] [Pubmed]
- Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(17):6275.
[Crossref] [Google Scholar] [PubMed]
- Hayatullina Z, Muhammad N, Mohamed N, Soelaiman IN. Virgin coconut oil supplementation prevents bone loss in osteoporosis rat model. Evid Based Complement Alternat Med. 2012;2012:237236.
[Crossref] [Google Scholar] [Pubmed]
- Hegazi R, El-Gamal M, Abdel-Hady N, Hamdy O. Epidemiology of and risk factors for type 2 diabetes in Egypt. Ann Glob Health. 2015;81(6):814-820.
[Crossref] [Google Scholar] [Pubmed]
- Griz LH, Bandeira F, Gabbay MA, Dib SA, de-Carvalho EF. Vitamin D and diabetes mellitus: An update 2013. Arq Bras Endocrinol Metabol. 2014;58(1):1-8.
[Crossref] [Google Scholar] [Pubmed]
- Hilger J, Friedel A, Herr R, Rausch T, Roos F, Wahl DA, et al. A systematic review of vitamin D status in populations worldwide. Br J Nutr. 2014;111(1):23-45.
[Crossref] [Google Scholar] [Pubmed]
- Holick MF. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin Proc. 2006;81:(3):353-373.
[Crossref] [Google Scholar] [Pubmed]
Citation: El-Desouki NI, Salem ML, Afifi DF, Nasef M, Abdallah FM (2023) The Helpful Effect of Vitamin D and Coconut Oil in Modulating the Histological Disorders of the Splenic Tissue in Hyperglycemic Mice. J Microb Biochem Technol. 15:548.
Copyright: © 2023 El-Desouki NI, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.