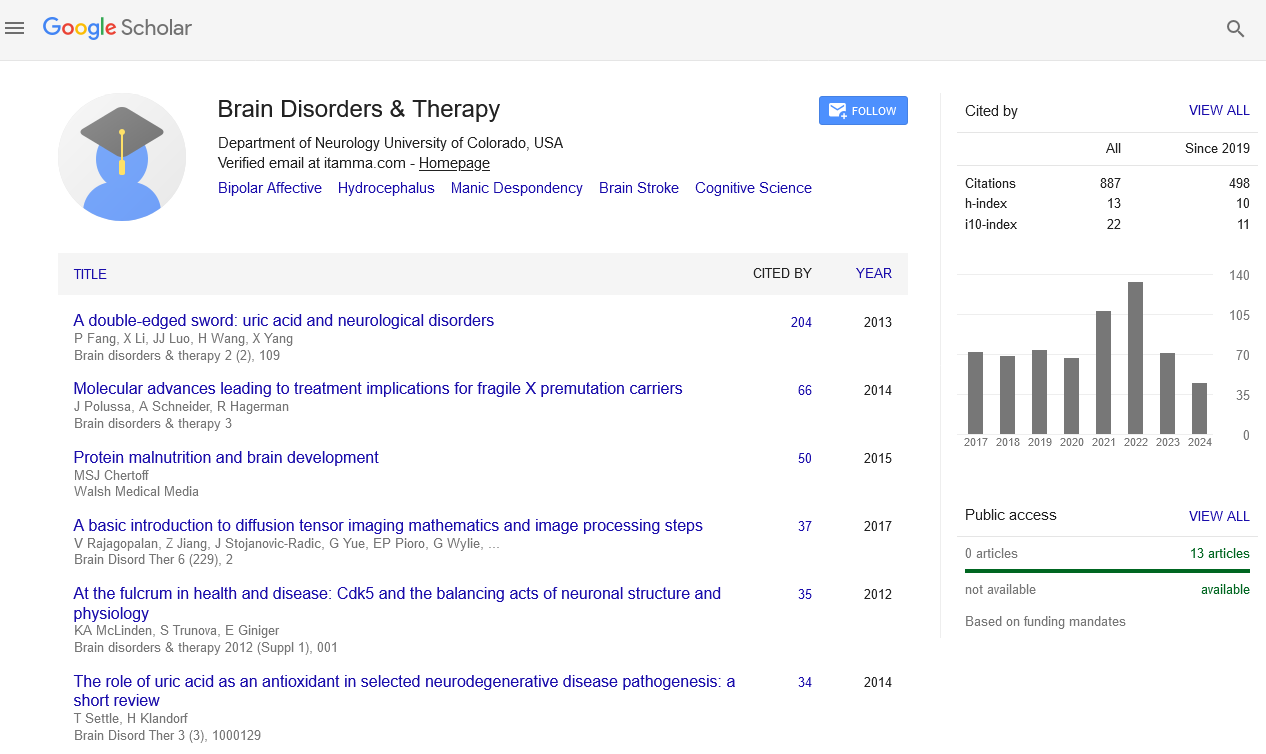
Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2022) Volume 0, Issue 0
Structure and Malformation of Pathology of Pituitary Metastasis of Breast Cancer : a systematic review
Ali Reza Arabestanino1,2,5*, Sina N. Irvani1,2, Arman Ai3, Bita Dinarvand4 and Parisa Aziminejadan42Shahid Beheshti University Medical of Science, Tehran, Iran
3Tehran University Medical of Science, Tehran, Iran
4Iran University Medical of Science, Tehran, Iran
5Sheikh Shakhbout Medical City, Abu Dhabi, United Arab Emirates
Received: 15-Nov-2022, Manuscript No. BDT-22-18772; Editor assigned: 23-Nov-2022, Pre QC No. BDT-22-18772 (PQ); Reviewed: 30-Nov-2022, QC No. BDT-22-18772; Revised: 07-Dec-2022, Manuscript No. BDT-22-18772 (R); Published: 16-Dec-2022, DOI: 10.35248/2168-975X.22.S7.178
Abstract
This study aims to systematically review the pituitary gland metastatic on breast cancer. A systematic review was conducted that follows the Preferential Reporting Guidelines for Systematic Review and Meta-Analysis (PRISMA). PubMed, EMBASE, Library Genesis were used to search for metastatic pituitary gland with an emphasis on breast cancer, which were released by June 2020. The terms “Pituitary Metastasis” and “Breast Cancer” were used to search for the publications. A total of 13 articles were included in this systematic review. Pituitary gland metastasis is rare and difficult to differentiate from without a pathologic diagnosis. Pituitary metastasis is very common in breast cancer and has rare manifestations. At the time of pituitary metastasis, most of the patients have clinical and radiological evidence of the disease. In the foreseeable future, play better methods and treatments for metastatic pituitary management on breast cancer in the age of precision medicine.
Keywords
Pituitary gland; Breast Cancer; Neoplasms; Pituitary carcinomas
INTRODUCTION
Metastases of the pituitary gland are extremely rare and represent only 1% of pituitary tumor lesions, in most cases the primary is of mammary or pulmonary origin [1,2]. At the time of diagnosis, most patients are elderly and present with advanced cancer disease with multiple metastatic sites [3]. Pituitary metastases can also be the first manifestation of occult primary cancer or the only metastatic site [4]. The role of pituitary hormones like prolactin, growth hormone, growth hormone releasing hormone, gonadotropin releasing hormone in breast cancer have been studied to a certain extent but not as exhaustively as estrogen. The pituitary hormones and their receptors could play a significant therapeutic role in breast cancer by themselves or in combination with the current standard treatment modalities. In the past decade growth hormone has been gaining a lot of attention in cancer development, progression and metastasis. Here we try to summarize the role of pituitary Metastasis in breast cancer with the available literature. The potential limitations and future directions of are also discussed in this systematic review.
Epidemiology
In generally although pituitary adenomas have been found by routine autopsy study to be surprisingly common, their emergence as clinical problems is far less frequent. Pituitary tumors are the third most common brain tumor in adults, accounting for 15% of adult tumors. But today, around the world, the number of patients with pituitary metastasis who also have breast cancer is very high. There is substantial variation in breast cancer rates among different countries. Rates are some six times higher in the USA, Canada, and northern Europe than in Asia or among black populations in Africa. These international differences in breast cancer rates do not appear to be determined primarily by variation in genetic susceptibility. Systematic surveys of pituitaries of patients dying of various causes have shown an incidence as high as 22.6% (some with multiple tumors) and as low as 8.8% [5,6]. Virtually all these tumors were unsuspected during life, although it must be emphasized that appropriate diagnostic studies were not carried out in these patients during life. The incidence increased with age. With advancements in screening, imaging, and therapeutics, cancer patients are enjoying improved survival and quality of life. Brain Metastases (BMs), Including pituitary metastasis with breast cancer, however, continue to be a major source of morbidity and mortality for nearly one fifth of adult cancer patients. The CBTRUS estimates that 11,000 new pituitary tumors will be diagnosed in the United States in 2015, with an incidence of 3.47 per 100,000. Most pituitary tumors are benign adenomas, although craniopharyngiomas and other types of histology comprise a minority of pituitary tumors [7]. Although pituitary adenomas are often cured by surgical resection, recurrences occur in 7% to 12%, and 60% to 65% of patients progress after subtotal resection; however, recurrences can be treated effectively with repeat resection or radiotherapy [8,9].
Methodology
This systematic review was performed to evaluate pituitary metastasis of breast cancer. The following are the inclusion and exclusion criteria.
Eligibility criteria
Studies were included if (1) exclusively targeted breast cancer patients; (2) published between 2017 and June 2020; (3) written in the English language; a systematic review was conducted that follows the preferential reporting guidelines for systematic review and meta-analysis (PRISMA). (4) published in a peer-reviewed journals indexed in PubMed, EMBASE, Library Genesis were used to search for metastatic pituitary gland with an emphasis on breast cancer; (5) they used any type of Pituitary Metastasis as a part of or the whole intervention; and (6) utilized a quantitative design for evaluation. Studies published in the past three years were included because a previous systematic review covered studies published before 2017. Studies which did not meet any of the aforementioned criteria were excluded. The studies were extracted independently by two authors. Duplicates were excluded by reference manager software (Endnote). We descriptively summarized the included articles with six tables. Additionally, studies were excluded if (1) they were incomplete or ongoing; (2) used qualitative methodology; (3) they were abstracts of conference proceedings, duplicates and letters to the editor, editorials, and commentaries.
Search strategy for identification of studies
This review followed internationally established Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) protocol guidelines (Moher et al.) for a thorough review and appropriate analysis of the peer reviewed articles for an authentic review article. In order to capture potentially relevant literature, systematic searches were performed on four electronic databases mentioned earlier for peer-reviewed articles that studied the Pituitary Metastasis of Breast Cancer patients. The following Boolean operators and combination of Pituitary Metastasis, breast cancer and intervention or programs search terms were used in the literature search process: “Pituitary Metastasis and Breast Cancer and Intervention or Program”. A total of 246 studies were identified through searches in electronic bibliographic databases. After removing duplicate publications from the obtained articles, 67 titles and abstracts were reviewed based on predetermined inclusion and exclusion criteria. This resulted in 40 papers that were considered for full-text review, which yielded 13 studies for inclusion in this systematic review. Three reviewers independently executed each above-mentioned step of the literature search process. If any discrepancies occurred between the reviewers, they were resolved through discussion and present the literature search procedure. The titles and abstracts of the articles were reviewed to see whether they meet the inclusion criteria or exclusion criteria. The articles that did meet the criteria were fully reviewed for data extraction (Figure 1).

Figure 1: PRISMA diagram depicting the literature search process.
Results
A total of 13 studies met the inclusion criteria for the review. Provides the summary of data extracted from the reviewed studies. The items in the table included information on the following characteristics (Table 1).
| Age (Y) | Most common histology | Second most common histology | Third most common histology |
|---|---|---|---|
| 15-19 | Tumors of the pituitary | Pilocytic astrocytoma | Neuronal and glioneuronal tumors |
| 20-34 | Tumors of the pituitary | Meningioma | Nerve sheath tumors |
| 35-44 | Meningioma | Tumors of the pituitary | Nerve sheath tumors |
| 45-54 | Meningioma | Tumors of the pituitary | Glioblastoma |
| 55-64 | Meningioma | Glioblastoma | Tumors of the pituitary |
| 65-74 | Meningioma | Glioblastoma | Tumors of the pituitary |
| 75-85 | Meningioma | Glioblastoma | Tumors of the pituitary |
| 85 | Meningioma | Glioblastoma | Tumors of the pituitary |
| Overall | Meningioma (7.61 per 100,000) | Tumors of the pituitary (3.29 per 100,000) | Glioblastoma (3.19 per 100,000) |
Table 1: Most common histology by age group. Adapted from [41].
Outcome measurements of the criteria recorded from participants included efficacy, metastatic effect of pituitary gland on participants’ breast cancer, which varied from study to study.
Identification tumors of pituitary
Pituitary adenomas are the most common tumors of the Sella and are manifest by either symptoms of mass effect or symptoms of hormone overproduction or underproduction. The histological and functional classification of pituitary adenomas is reviewed in relation to their clinical presentation and potential therapies [10]. Most pituitary adenomas are slow-growing tumors, but some have a higher growth rate and can be invasive. Pituitary carcinomas are very rare and are defined by the presence of metastasis or cerebrospinal fluid dissemination [11]. Pituitary adenomas are clinically significant because they may present with symptoms of mass effect, especially visual compromise, and symptoms of endocrine dysfunction. Pituitary adenomas have been classified in various ways.
The latest Endocrine Tumors Classification of the WHO includes a pituitary adenoma classification based on the previous morpho functional classification, but it gives more importance to the classical types of adenomas with clear definitions, providing an easier approach to these tumors. The remaining pathologic categories are included, but at a secondary level as microscopic variants (Table 2).
| Tumors of pituitary | Tumors of endocrine organs |
|---|---|
| Pituitary adenomas | 1. PRL-producing tumors |
| 2. Growth hormone-producing tumors | |
| 3. Adrenocorticotropin-producing tumor | |
| 4. Gonadotropin-producing tumor | |
| 5. Thyrotropin producing adenomas | |
| 6. Null cell adenomas | |
| 7. Plurinominal adenomas | |
| Pituitary carcinoma | |
| Gangliocytoma | |
| Mesenchymal tumors | 1. Chordoma |
| 2. Meningioma | |
| Granular cell tumors | |
| Secondary tumors |
Table 2: Tumors of pituitary, source: Tumors of endocrine organs, WHO classification (2004).
Discussion
In most of the studies on metastatic involvement of the pituitary gland, breast and lung cancers were the most primary tumors comprising approximately two-thirds of cases, but metastasis from lymphoma, leukemia, melanoma, kidney, colon, and prostatic cancer were also reported [10].
There is a strong correlation between the occurrence of pituitary metastasis and the presence of widespread, especially skeletal, dissemination of metastatic tumor. This suggests that the appearance of pituitary metastasis represents extensive disease. Rarely, a patient with no previous history of malignancy presents with symptoms consistent with pituitary adenoma [11,12]. Occasionally, symptoms produced by pituitary metastasis occur in patients with no prior history of malignancy. In the 60 reported cases of symptomatic pituitary metastasis found in the literature, (30%) had no known malignancy. This percentage is probably artificially high since this type of presentation is unusual and more likely to be reported [3,12-14]. The current literature on pituitary metastases in breast cancer generally describes lesions discovered by neurosurgeons and general surgeons. The present study provides an integrated systematic review of patients who report pituitary metastasis to breast cancer. Therefore, the present study aimed to describe and treat how to diagnose a patient with pituitary metastasis who is suspected of breast cancer. Between a long-term period until June 2020, they were considered for use in the present study.
The symptoms produced by metastatic pituitary involvement are shown. Diabetes insipidus was the presenting symptom in 70% of the patients. This finding reflects the propensity for pituitary metastasis to localize in the posterior lobe of the pituitary. In cases of pituitary metastasis where both lobes were examined, over 80% had involvement of the posterior lobe [12,13].
The treatment of patients harboring pituitary metastasis depends on the symptoms produced by the metastasis, as well as the extent of their systemic neoplastic disease. Obviously, every attempt should be made to preserve visual function, even in patients with extensive disease. This usually can be accomplished by trans-sphenoidal decompression followed by radiation therapy (Figure 1).
Surgery also should be considered in cases where accurate histological diagnosis would affect future treatment.
i. Histopathological Pituitary Metastasis (HPM)
ii. Histopathological Breast Cancer (HBC)
Histopathological Pituitary Metastasis (HPM)
Modern and general concepts of pathophysiological and therapeutic treatment of pituitary metastasis are significantly different from the concepts that existed from the time of Harvey Cushing until just a few years ago. Because traditional concepts have dominated our thinking for a long time, reviewing them is practical and useful and is better understood and understood for new therapies [14].
Pituitary gland adenoma accounts for 20% of all central nervous system tumors in the adult population [15]. The percentage of pituitary tumors that are benign and primary carcinoma only account for 0.1%-0.2% of all cases [16], Metastasis to the pituitary gland is unusual. Therefore, the pituitary gland is not a common site of metastasis. Pituitary metastases were found in 1%-3.6% of patients with malignant tumors [17]. Pituitary metastases occur most commonly in the elderly, especially those aged 60-70 years [1].
Pituitary adenoma is the most common cause of pituitary masses, constituting approximately 10% of all intracranial neoplasms. Pituitary gland metastasis accounts for only approximately 1% of all pituitary tumors in previous serial studies [18].
Breast tumors and lung tumors are the most common primary sites of malignant metastases to the pituitary gland, but other sites such as the gastrointestinal tract, prostate, kidney, thyroid, and pancreas have also been reported [2]. Morita et al. reported metastases from breast cancer accounted for 33% of pituitary metastases and those from the lung accounted for 36% in 36 clinically symptomatic patients [19].
In breast cancer patients, the development of metastatic disease is considered to be the major cause of mortality [20]. The invasion- metastasis cascade is a multistep process in which breast cancer cells invade the systemic circulation and distant tissues, and cause the metastatic colonization of different organs, including the bones, lungs and central nervous system [21]. Being highly vascularized, the pituitary gland is a target structure for metastatic spread, accounting for 0.14%-28% of all reported brain metastases following autopsy [22]. In total, 6%-8% of breast cancer cases demonstrate metastatic spread to the pituitary gland [23].
Electron microscopy: granules of the cells pituitary
Normal pituitary gland cells can be classified according to their size and density, as seen under an electron microscope: Gray found that normal gland cells can be divided into six types based on the size of the granules. Their cytoplasm contains cells with granules 100 nm to 160 nm in diameter that contain TSH. Granules with a diameter of 200 nm-160 nm were thought to contain LH. Granules with a diameter of 200 nm-270 nm were considered characteristic of FSH, and those with a diameter of 270-410 were correlated with ACTH. The granules of classical acidophiles were larger: 410 nm-500 nm for GH and 500 nm-600 nm for prolactin. The identification of granules by parallel display of hormones was not confirmed by immunological techniques. In addition, differences in granule size are more pronounced in resting cells. In active secretory cells, the size of the granule decreases and is not a reliable criterion for identifying the hormone produced (sometimes it is of little use in the absence of the necessary access) [24,25]. Some pituitary adenomas have granules that are easily recognized as GH21 or prolactin (Table 3) [26].
| Types | Kovacs et al. | Landolt |
|---|---|---|
| Prolactinoma | 25 | 39 |
| GH | 22 | 33 |
| Stem cell | 5 | 5 |
| ACTH | 14 | 4 |
| Mixed | 5 | N/A |
| TSH | 1 | N/A |
| Gonadotropic | N/A | N/A |
| Undifferentiated | 26 | 24* |
Note: Endocrine-inactive and oncocytoma.
Table 3: Relative of various-types of pituitary adenomas.
Histopathological Breast Cancer (HBC)
Breast cancer is the most common cancer of women worldwide [27]. Breast cancer accounts for 22% of all female cancers, which is more than twice the occurrence of cancer in women at any other [28]. Male breast cancer is rare compared with female breast cancer. Female: male incidence ratios vary from 70 to 130 around the world.
However, in histopathological practice, cell type characteristics, number of cells, type and location of secretion, immunohistochemical profile and architectural characteristics determine if the tumor is ductal or lobular, in addition to its sub-classifications, rather than its precise location in the mammary tissue [29,30]. About 50% to 80% of newly diagnosed breast cancer cases are called Invasive Ductal Carcinoma (IDC); the rest of the cases are classified as Invasive Lobular Carcinoma (ILC) [31].
IDCs can be classified as “no specific type” because these tumors do not present sufficient morphological characteristics to be determined as a characteristic histological type; they can also be recognized as a “special type” if they present sufficient distinctive characteristics, and particular cellular and molecular behavior [32,33]. common special types of breast cancer include: medullary carcinoma, metaplastic carcinoma, apocrine carcinoma, mucinous carcinoma, cribriform carcinoma, tubular carcinoma, neuroendocrine carcinoma, classic lobular carcinoma, and pleomorphic lobular carcinoma (Table 4) [34].
| Risk factors |
|---|
| Early menarche |
| Late menopause |
| Obesity (postmenopausal women) |
| Oestrogen replacement therapy |
| Older age at first full-tem birth |
| Nulliparity |
| Oral contraceptives |
Table 4: Breast cancer risk factors.
A relatively large percentage of the samples that are analyzed in pathology labs are from breast cancer patients, since this disease is the most prevalent form of cancer among women [16]. Analysis methods that are routinely performed by pathologists, such as determination of the histological grade and the hormone receptor status by Immuno Histo Chemistry (IHC), can be tedious and are hampered by observer variability [3,17]. The histological tumor grade is commonly determined according to the modified Bloom– Richardson system, which consists of semiquantitative assessment of nuclear atypia, tubule formation, and mitotic activity [34,35]. The analysis of immunohistochemically stained slides mainly involves the estimation of the number of cells that are positive for a particular antigen and the degree of positivity (staining intensity) [36,37]. Pathology labs are currently undergoing a transformation toward a fully digital workflow [14]. In addition to the digital management of tissue samples, pathology orders, and reports, this includes the digitization of histopathology slides and use of computer monitors for viewing them, which aims to replace the optical microscope as the primary tool used by pathologists. This transformation has only recently been enabled by the introduction of cost and time efficient Whole Slide Imaging (WSI) scanners, as successors to microscope-mounted digital cameras. This process of adoption of digital slide images is somewhat analogous to the digitization of radiological imaging. However, a full analogy is difficult to establish because in pathology the primary object of analysis is the tissue rather than the image [3].
One of the main benefits of digital slides compared to conventional glass slides is that they enable the use of quantitative automatic image analysis methods. These methods have the potential to tackle the problems that stem from the subjective interpretation by pathologists and, at the same time, reduce their workload [17]. However, some techniques developed for other tissue types or microscopy modalities that are relevant for the scope of our paper are mentioned throughout the text when appropriate. For a broader overview of digital pathology and the use of automatic methods for analysis of histopathology slides, we refer the reader to the recent reviews [38-40].
Pituitary metastases are most prevalent in the sixth and seventh decades of life [3]. There is no consensus on improving survival rate with surgical resection (sometimes in some cases superficial and immediate treatment has been mentioned, but there is no average of all studies to improve survival rate with surgical removal), but there is improvement in symptoms by local decompression of the tumor, although it requires a combination of different therapies [41-44].
The prognosis is bad, not because of its location, but because of the aggressiveness of the tumors. The median survival in clinical series was 6-7 months according to the available evidence, but among the studies performed by Netunga Pono and Sarein identified only 10% of survivors a year later, with a maximum survival of 3 years. Survival can be improved when the pituitary lesion is a single metastasis [25,45-48].
Conclusion
Although the pituitary gland is a rare site of metastasis, pituitary metastases should be examined in the face of any clinical or biochemical hormonal abnormalities. Lesions and signs of a pituitary lesion indicate metastatic disease, even in patients with no known onset and its clinical symptoms and MRI findings are similar to those of other sellar tumors, its progression is rapid, and the prognosis is poor. Our case emphasizes. There is no consensus on how to improve survival rates with surgical resection (in some cases, superficial and immediate treatment has been mentioned, but there is no overall average to improve survival rates with surgical removal) the importance of a differential diagnosis of invasive sellar lesions, particularly in people over the age of 60 who have diabetes insipidus.
Disclosure
Conflict of interest the authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
References
- Komninos J, Vlassopoulou V, Protopapa D, Korfias S, Kontogeorgos G, Sakas DE, et al. Tumors metastatic to the pituitary gland: Case report and literature review. J Clin Endocr.2004;89(2):574-580.
[Crossref] [Google Scholar] [PubMed]
- Spinelli GP, Lo Russo G, Miele E, Prinzi N, Tomao F, Antonelli M, et al. Breast cancer metastatic to the pituitary gland: A case report. World J Surg Oncol. 2012;10(1):1-4.
[Crossref] [Google Scholar] [PubMed]
- Max MB, Deck MD, Rottenberg DA. Pituitary metastasis: Incidence in cancer patients and clinical differentiation from pituitary adenorna. Neurology. 1981;31(8):998-1002.
[Crossref] [Google Scholar] [PubMed]
- Ntyonga-Pono MP, Thomopoulos P, Luton JP. Pituitary metastases. 3 cases. Presse Med.1999;28(29):1567-1571.
[Google Scholar] [PubMed]
- Costello RT. Subclinical adenoma of the pituitary gland. Am J Pathol.1936;12(2):205.
[Google Scholar] [PubMed]
- McCormick WF, Halmi NS. Absence of chromophobe adenomas from a large series of pituitary tumors. Arch Pathol. 1971;92(4):231-238.
[Crossref] [Google Scholar] [PubMed]
- Ezzat S, Asa SL, Couldwell WT, Barr CE, Dodge WE, Vance ML, et al. The prevalence of pituitary adenomas: A systematic review.Cancer.2004;101(3):613-619.
[Crossref] [Google Scholar] [PubMed]
- Dallapiazza RF, Grober Y, Starke RM, Laws Jr ER, Jane Jr JA. Long-term results of endonasal endoscopic transsphenoidal resection of nonfunctioning pituitary macroadenomas. Neurosurgery. 2015;76(1):42-53.
- Berkmann S, Schlaffer S, Nimsky C, Fahlbusch R, Buchfelder M. Follow-up and long-term outcome of nonfunctioning pituitary adenoma operated by transsphenoidal surgery with intraoperative high-field magnetic resonance imaging. Acta Neurochir. 2014;156(12):2233-2243.
- Kurkjian C, Armor JF, Kamble R, Ozer H, Kharfan-Dabaja MA. Symptomatic metastases to the pituitary infundibulum resulting from primary breast cancer. Int J Clin Oncol. 2005;10(3):191-194.
[Crossref] [Google Scholar] [PubMed]
- Chaudhuri R, Twelves C, Cox TC, Bingham JB. MRI in diabetes insipidus due to metastatic breast carcinoma. Clin Radiol. 1992;46(3):184-188.
[Crossref] [Google Scholar] [PubMed]
- Hagerstrand I, Schoneback J. Metastases to the pituitary gland. Acta Pathol Microbiol Scand. 1969;75:64-70.
[Crossref] [Google Scholar] [PubMed]
- Duchen LW. Metastatic carcinoma in the pituitary gland and hypothalamus. J Pathol Bacterial.1966;91(2):347-355.
[Crossref] [Google Scholar] [PubMed]
- Roessmann U, Kaufman B, Friede RL. Metastatic lesions in the sella turcica and pituitary gland. Cancer. 1970;25(2):478-480.
[Crossref] [Google Scholar] [PubMed]
- Teears RJ, Silverman EM. Clinicopathologic review of 88 cases of carcinoma metastatic to the pituitary gland. Cancer. 1975;36(1):216-220.
[Crossref] [Google Scholar] [PubMed]
- Cushing H. The pituitary body and its disorders: Clinical states produced by disorders of the hypophysis cerebri. jB Lippincott; 1912.
- Daly AF, Rixhon M, Adam C, Dempegioti A, Tichomirowa MA, Beckers A. High prevalence of pituitary adenomas: A cross-sectional study in the province of Liege, Belgium. J Clin Endocrinol Metab. 2006;91(12):4769-4775.
[Crossref] [Google Scholar] [PubMed]
- Pichard C, Gerber S, Laloi M, Kujas M, Clemenceau S, Ponvert D, et al. Pituitary carcinoma: Report of an exceptional case and review of the literature. J Endocrinol Invest. 2002;25(1):65-72.
[Crossref] [Google Scholar] [PubMed]
- Scully RE, Mark EJ, McNeely WF. Case records of the Massachusetts general hospital. N Engl J Med. 2001;345(20):1483-1488. .
- Freda PU, Post KD. Differential diagnosis of sellar masses. Endocrinol Metab Clin North Am.1999;28(1):81-117.
- Morita A, Meyer FB, Laws ER. Symptomatic pituitary metastases. J Neurosurg. 1998;89(1):69-73 .
[Crossref] [Google Scholar] [PubMed]
- Gupta GP, Massagué J. Cancer metastasis: Building a framework. Cell. 2006;127(4):679-695.
[Crossref] [Google Scholar] [PubMed]
- Nguyen DX, Bos PD, Massagué J. Metastasis: From dissemination to organ-specific colonization. Nat Rev Cancer.2009;9(4):274-284.
[Crossref] [Google Scholar] [PubMed]
- Sioutos P, Yen V, Arbit E. Pituitary gland metastases. Ann Surg Oncol.1996;3(1):94-99.
[Crossref] [Google Scholar] [PubMed]
- DR F. Couldwell WT. Metastases to the pituitary gland. Neurosurg Focus. 2004;16:E8.
[Crossref] [Google Scholar] [PubMed]
- Gray AB. Analysis of diameters of human pituitary hormone secretory granules. Acta Endocrinol. 1977;85(2):249-255.
[Crossref] [Google Scholar] [PubMed]
- M. Herlant. Introduction. in: Ultrastructure in Biological Systems. Academic Press. New York. 1975;3-19.
- Peake GT, Mckeel DW, Jarett L, Daughaday WH. Ultrastructural, histologic and hormonal characterization of a prolactin-rich human pituitary tumor. J Clin Endocrinol. 1969;29(11):1383-1393.
[Crossref] [Google Scholar] [PubMed]
- Parkin DM, Stjernswärd J, Muir CS. Estimates of the worldwide frequency of twelve major cancers. Bull World Health Org. 1984;62(2):163-182.
[Crossref] [Google Scholar] [PubMed]
- Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94(2):153-156.
[Crossref] [Google Scholar] [PubMed]
- Makki J. Diversity of breast carcinoma: Histological subtypes and clinical relevance. Clin Med Insights Pathol. 2015;8(1):23- 31.
[Crossref] [Google Scholar] [PubMed]
- Nounou MI, ElAmrawy F, Ahmed N, Abdelraouf K, Goda S, Syed-Sha-Qhattal H. Breast cancer: Conventional diagnosis and treatment modalities and recent patents and technologies.Breast Cancer Basic Clin Res. 2015;9:17-34.
[Crossref] [Google Scholar] [PubMed]
- Henry NL, Cannon‐Albright LA. Breast cancer histologic subtypes show excess familial clustering. Cancer. 2019;125(18):3131-3138.
[Crossref] [Google Scholar] [PubMed]
- Masood S. Breast cancer subtypes: Morphologic and biologic characterization. Women’s Health. 2016;12(1):103-119.
[Crossref] [Google Scholar] [PubMed]
- Branch JR CL, Laws JR ER. Metastatic tumors of the sella turcica masquerading as primary pituitary tumors. J Clin Endocrin Metab. 1987;65(3):469-474.
[Crossref] [Google Scholar] [PubMed]
- Houck WA, Olson KB, Horton J. Clinical features of tumor metastasis to the pituitary. Cancer. 1970;26(3):656-659.
[Crossref] [Google Scholar] [PubMed]
- Kimmel DW, O'Neill BP. Systemic cancer presenting as diabetes insipidus. Clinical and radiographic features of 11 patients with a review of metastatic‐induced diabetes insipidus. Cancer. 1983;52(12):2355-2358.
[Crossref] [Google Scholar] [PubMed]
- Weil RJ. Pituitary metastasis. Arch Neurol.2002;59:1962-1963.
- Duchen LW. Metastatic carcinoma in the pituitary gland and hypothalamus. J Pathol Bacterial.1966;91(2):347-355.
[Crossref] [Google Scholar] [PubMed]
- Yap HY, Tashima CK, Blumenschein GR, Eckles N. Diabetes insipidus and breast cancer. Arch Intern Med. 1979;139(9):1009-1011.
[Crossref] [Google Scholar] [PubMed]
- Ostrum QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, et al. CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol. 2014;16(S4):1-63.
[Crossref] [Google Scholar] [PubMed]
- Landolt AM. Progress in Pituitary Adenoma Biology Results of Research and Clinical Applications. Adv Tech Stand Neurosurg. 1978;3-49.
- Sturm I, Kirschke S, Krahl D, orken B. Panhypopituitarism in a patient with breast cancer. Onkologie. 2004; 27: 280-282.
- DeSantis CE, Siegel RL, Sauer AG, Miller KD, Fedewa SA, Alcaraz KI, et al. Cancer statistics for African Americans, 2016: Progress and opportunities in reducing racial disparities. CA: Cancer J Clin. 2016;66(4):290-308.
[Crossref] [Google Scholar] [PubMed]
- Jones SL, Hadjistavropoulos HD, Gullickson K. Understanding health anxiety following breast cancer diagnosis. Psychol Health Med . 2014;19(5):525-535.
[Crossref] [Google Scholar] [PubMed]
- Kleinberg DL. Early mammary development: growth hormone and IGF-1. J Mammary Gland Biol Neoplasia. 1997;2(1):49-57.
[Crossref] [Google Scholar] [PubMed]
- Sternlicht MD. Key stages in mammary gland development: The cues that regulate ductal branching morphogenesis. Breast Cancer Res. 2005;8(1):1-1.
[Crossref] [Google Scholar] [PubMed]
- Gopan T, Toms SA, Prayson RA, Suh JH, Hamrahian AH, Weil RJ. Symptomatic pituitary metastases from renal cell carcinoma. Pituitary. 2007;10(3):251-259.
[Crossref] [Google Scholar] [PubMed]
Citation: Arabestanino AR, Irvani S N, Ai A, Dinarvand B, Aziminejadan P (2022) Structure and Malformation of Pathology of Pituitary Metastasis of Breast Cancer: A Systematic Review. Brain Disord Ther. S7:178.
Copyright: © 2022 Arabestanino AR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.