Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Review Article - (2025) Volume 16, Issue 6
Restrictive vs. Liberal Transfusion Strategy in Patients with Myocardial Infarction and Anaemia: A Systematic Review and Meta-Analysis
Mazin Ali1, Sreenidhi Pithani1 and Denise E. Jackson1,2*2Department of Thrombosis and Vascular Diseases Laboratory, School of Health and Biomedical Sciences,, Australia
Received: 18-Dec-2025, Manuscript No. JBDT-25-30681; Editor assigned: 22-Dec-2025, Pre QC No. JBDT-25-30681 (PQ); Reviewed: 05-Jan-2026, QC No. JBDT-25-30681; Revised: 12-Jan-2026, Manuscript No. JBDT-25-30681 (R); Published: 19-Jan-2026, DOI: 10.4172/2155-9864.25.16.636
Abstract
References
Best Betting Sites in Turkey Blog | Mexican Food Recipes Casino Sites in Argentina Best Betting Sites in Canada Blog | Turkish Food Recipes Fan Tokens Info Blog | Vietnamese Food Recipes Engine Technologies Articles Blog | Yacht Charter in UAE All About Digital Coins Blog | Yacht Charter in Balkans Casino Sites in Bangladesh Blog | Yacht Charter in Australia Mortgage info in New York Blog | Yacht Charter in China Best Betting Sites in Nigeria Blog | Yacht Charter in Spain Best Betting Sites in United Arab Emirates Blog | Yacht Charter in France Places to Visit in ChinaKeywords
Restrictive transfusion; Liberal transfusion; Myocardial infarction; Anaemia; Red blood cell transfusion, Meta-analysis
Introduction
Pathophysiological rationale for transfusion
The rationale for transfusion during MI is rooted in the fundamental relationship between haemoglobin concentration and oxygen delivery [1-4]. In anaemic states, reduced oxygen content in arterial blood leads to compensatory increases in heart rate, stroke volume, and oxygen extraction at the tissue level [5,6]. While these mechanisms preserve tissue oxygenation in healthy individuals, they are inadequate in ischaemic myocardium, where coronary flow reserve is already limited. Therefore, even small reductions in haemoglobin can aggravate myocardial hypoxia, which may precipitate further infarction and ventricular dysfunction [7].
Transfusing RBCs to increase haemoglobin theoretically improves oxygen carrying capacity and reduces the metabolic stress imposed by anaemia. Yet, excessive transfusion may increase blood viscosity and left ventricular afterload, which compromises coronary perfusion pressure and oxygen diffusion across the capillary bed [8,9]. Moreover, stored red cells lose membrane deformability and nitric oxide bioavailability over time, causing impairment of the microcirculatory flow and promoting vasoconstriction [10]. Observational studies have also linked liberal transfusion to increased mortality, possibly mediated by inflammatory activation and oxidative injury [11].
These competing physiological mechanisms underscore why neither a universally restrictive nor a universally liberal transfusion threshold can be presumed safe without empirical validation. The ideal approach likely depends on balancing oxygen delivery and rheological factors in the infarcted heart. This interplay forms the foundation for current research and meta-analyses evaluating whether restrictive thresholds, transfusing only when haemoglobin falls below 7-8 g/dL, can safely replace traditional liberal thresholds of 9-10 g/dL in anaemic MI patients.
Evolution of transfusion evidence
Historically, clinicians adhered to the “10/30 rule,” which meant transfusing when haemoglobin dropped below 10 g/dL or haematocrit below 30%. However, this convention was challenged by the landmark TRICC trial, which demonstrated that in critically ill adults, a restrictive transfusion strategy (Hb<7 g/dL) was as safe as a liberal one (Hb<10 g/dL) [6]. The 2021 Cochrane systematic review later helped reinforce these findings, showing across 48 randomised controlled trials involving over 21,000 participants that restrictive strategies reduced risk of transfusion by up to 43% without increasing 30-day mortality [12].
Despite these advances, patients with active myocardial ischaemia were largely excluded from early studies, leaving clinicians uncertain whether restrictive thresholds are appropriate in the context of acute coronary syndromes. Given the myocardium’s high oxygen demand and limited tolerance for hypoxia, there has been longstanding concern that restrictive transfusion could exacerbate ischaemic injury in MI [13]. This uncertainty catalysed several Randomised Controlled Trials (RCTs) dedicated to anaemic MI populations over the past decade.
Major randomised controlled trials in MI
Evidence in MI-specific settings now draws on six pivotal RCTs. The CRIT Pilot Study (Cooper, et al. 2011) randomised 45 patients with acute MI and anaemia to restrictive (Hb ≤ 8 g/dL) versus liberal (Hb ≤ 10 g/dL) transfusion. Although underpowered, it showed a trend toward higher reinfarction and death in the liberal group [14].
The Restrictive and Liberal Transfusion Strategies in Patients with Acute Myocrdial Infarction (REALITY) trial expanded on this foundation, enrolling 668 MI patients with haemoglobin 7-10 g/dL. It demonstrated that restrictive transfusion was non-inferior to liberal transfusion for 30-day Major Adverse Caediovascular Events (MACE) [15]. The REALITY one-year extension confirmed the absence of excess mortality or recurrent MI with restrictive thresholds [16]. The Myocardial Ishemia and Transfusion (MINT) trial, largest to date (n=3,504), found no significant difference in 30-day mortality or recurrent infarction between restrictive (≤ 8 g/dL) and liberal (≤ 10 g/dL) thresholds, cementing its short-term safety. Follow-up data from Simon et al. (2024) extended these findings to six months, showing no long-term detriment with restrictive transfusion [17]. Finally, DeFilippis, et al. demonstrated consistent safety of restrictive strategies across both type 1 and type 2 MI, underscoring their applicability to varied pathophysiological subtypes [18]. Together, these trials form the empirical core for current systematic review and meta-analyses.
Current evidence and rationale for investigation
Pooled data from these RCTs consistently show that a restrictive transfusion threshold (7-8 g/dL) achieves comparable short-term outcomes to a liberal approach (9-10 g/dL) for 30-day mortality and MACE [17-21]. This supports the view that immediate transfusion at higher thresholds offers limited incremental benefit. Nevertheless, several key uncertainties persist. Longitudinal analyses of both REALITY and MINT observed subtle divergence in survival curves beyond 30 days, suggesting potential delayed effects of anaemia correction or post-transfusion physiology over six to twelve months [16,17]. Additionally, recurrent myocardial infarction, a common endpoint with major prognostic and resource implications, has not been uniformly reported across trials. While some data imply fewer reinfarctions with restrictive transfusion, possibly due to improved haemorheology and lower intravascular volume load, the consistency of this finding across studies remains unclear in light of all evidences [17,18].
Aim and research questions
The overarching aim of this systematic review and meta-analysis is to determine the optimal transfusion threshold in adults hospitalised with myocardial infarction and anaemia by comparing the clinical outcomes of restrictive and liberal red blood cell transfusion strategies. Drawing upon data from all six randomised controlled trials, this review seeks to clarify the short-term, medium-term, and recurrent ischaemic consequences of transfusion practice in this high-risk group.
Specifically, it aims to evaluate whether a restrictive transfusion threshold (haemoglobin ≤ 7-8 g/dL, target 8-10 g/dL) compared with a liberal threshold (haemoglobin ≤ 9-10 g/dL, target ≥ 10 g/dL):
• Achieves comparable or improved outcomes for 30-day Major Adverse Cardiovascular Events (MACE) and all-cause mortality,
• Maintains safety and clinical effectiveness over an extended 6-month to 1-year follow-up period, and
• Reduces the incidence of recurrent myocardial infarction compared with liberal transfusion.
By systematically addressing these three research questions, the meta-analyses aim to resolve key uncertainties and consolidate the available information in transfusion management for anaemic MI in order to provide evidence directly applicable to current clinical guidelines.
Methods
Search strategy
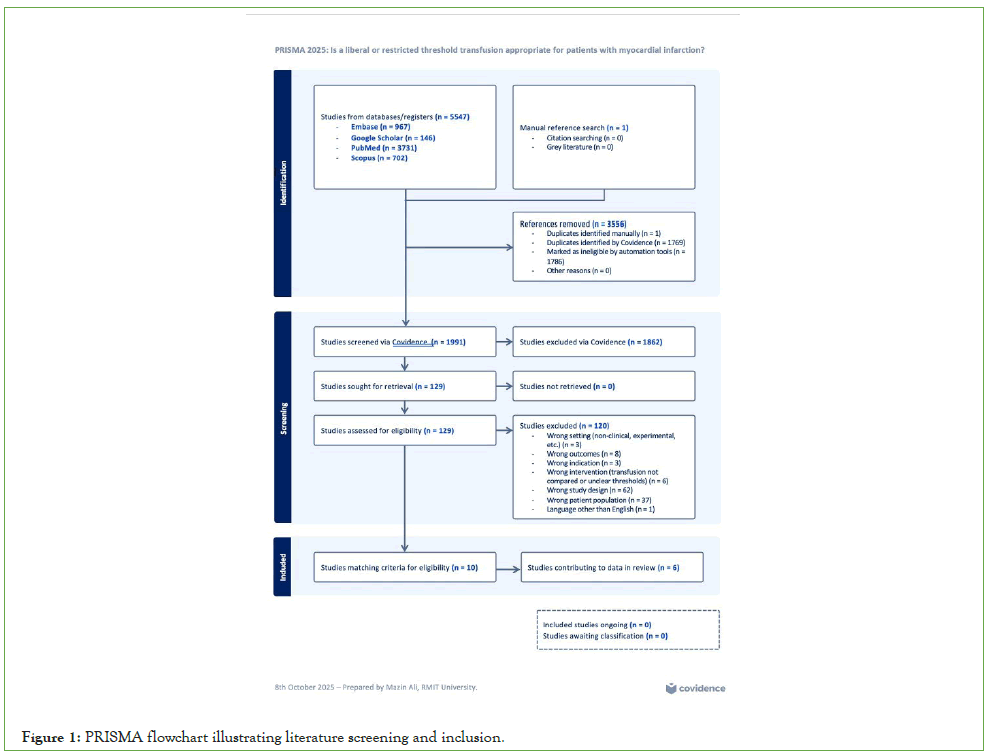
A systematic literature search was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines [19]. The search spanned five databases, PubMed, Embase, Scopus, Google Scholar, and the Cochrane Library, covering publications from January 2000 to March 2024. Medical Subject Headings (MeSH) and free-text terms relating to myocardial infarction, anaemia, blood transfusion, and transfusion thresholds were combined using Boolean operators (“AND” and “OR”) to maximise retrieval sensitivity. Searches were performed without geographical restrictions but were limited to English-language studies. All records were imported into the Covidence systematic review platform, where duplicates were removed automatically. A total of 5,547 records were identified, and 1,991 unique studies were retained for screening after duplicate removal.
Screening and eligibility
Titles and abstracts were screened independently by using covidence following PRISMA methodology [19]. Potentially eligible full-text articles were then assessed against predefined inclusion and exclusion criteria. Inclusion criteria comprised randomised controlled trials comparing restrictive versus liberal red blood cell transfusion strategies in adult patients with acute or recent myocardial infarction. Exclusion criteria included non-MI populations, non-comparative or observational designs, non-English publications, systematic reviews, subgroups, and paediatric cohorts. Following full-text review of 129 articles, six RCTs met all eligibility requirements and were included in the quantitative synthesis [13-17]. Reasons for study exclusion were documented, and the complete selection process is presented in the PRISMA flow diagram (Figure 1). The review protocol adhered to recommendations outlined in the Cochrane handbook for systematic reviews of interventions [20].
Figure 1: PRISMA flowchart illustrating literature screening and inclusion.
Data extraction
Data extraction was performed independently using a standardised proforma to ensure consistency. Extracted information included: Author, publication year, study design, sample size, transfusion thresholds, follow-up duration, and primary and secondary outcomes. Event counts for both liberal and restrictive groups were recorded and cross-verified with the original publications. Any discrepancies were resolved by consensus, and missing data were sought from corresponding authors when necessary. All data were initially organised in Microsoft Excel and subsequently imported into Review Manager 5.4 (RevMan; Cochrane collaboration, 2020) for meta-analysis and formation of standardised forest plots [20,21].
Data synthesis and statistical analysis
Quantitative synthesis was conducted using a random-effects model (DerSimonian and Laird method) to account for clinical and methodological heterogeneity between studies [22]. Effect measures were expressed as Risk Ratios (RRs) or Odds Ratios (ORs) with 95% Confidence Intervals (CIs). Heterogeneity was quantified using the I2 statistic, with values above 50% indicating substantial result variability [23].
Outcomes were analysed across three prespecified intervals. The short-term period (≤ 30 days) evaluated all-cause mortality and Major Adverse Cardiac Events (MACE), which reflected the acute effects of transfusion thresholds on immediate post-MI prognosis. The extended follow-up interval (6 months to 1 year) assessed longer-term mortality and composite cardiac events to explore sustained effects of transfusion strategies beyond hospitalisation. Lastly, recurrent myocardial infarction was treated as a distinct outcome to evaluate potential harm from excessive transfusion or oxygen-delivery imbalance after discharge. Pooled estimates were generated using inverse-variance weighting, and forest plots were produced in RevMan 5.4 to visualise effect sizes. Publication bias was evaluated visually through funnel-plot asymmetry [24]. A p value<0.05 was considered statistically significant.
All procedures conformed to Cochrane methodological standards and the 2023 AABB international transfusion guidelines, which ensured that evidence synthesis was robust, reproducible, and clinically relevant [9,20].
Results
Study selection and characteristics
A total of six Randomised Controlled Trials (RCTs) met inclusion criteria, encompassing 6,849 patients hospitalised with acute Myocardial Infarction (MI) and anaemia across North America, Europe, and multinational networks. These included the CRIT pilot study [14], REALITY trial [15], REALITY one-year follow-up [16], MINT Trial [25], MINT six-month extension [17], and evaluating transfusion thresholds in both type 1 and type 2 MI [13-17]. Collectively, these RCTs represent the highest-quality evidence comparing restrictive (transfusion at ≤ 7-8 g/dL, target 8-10 g/dL) versus liberal (transfusion at ≤ 10 g/dL, target ≥ 11 g/dL) Red Blood Cell (RBC) transfusion strategies in acute MI with anaemia.
Across studies, mean age was approximately 70 years, and women represented ~40% of participants. Study designs were largely prospective (five RCTs) with one retrospective multicentre analysis. Restrictive transfusion thresholds were defined as Hb ≤ 7-8 g/dL (target 8-10 g/dL), while liberal thresholds were ≤ 10 g/dL (target ≥ 11 g/dL). Follow-up durations ranged from 30 days to 12 months. Baseline comorbidities were similar between arms, although liberal cohorts tended to include slightly more patients with prior heart failure or renal impairment [13-17].
All trials reported adherence to good clinical practice, ethics approval, and >95 % follow-up completion. These characteristics are summarized, which outlines study design, population, and key parameters (Table 1).
| Study | Study design | Country | Study period | Sample size (n) | Parameters measured |
|---|---|---|---|---|---|
| Cooper, et al. 2011 [7] | Prospective | USA | 2007-2009 | 45 | Hb threshold <8 vs. <10 g/dL; Death/Reinfarction |
| Ducrocq, et al. 2021 [18] | Prospective | France/ Spain | 2018-2020 | 666 | Hb threshold <8 vs. <10 g/dL; MACE (30 days) |
| Carson, et al. 2023 [5] | Multicentre | USA/Canada/France | 2019-2021 | 3415 | Hb threshold ≤8 vs. ≤10 g/dL; Death/Recurrent MI |
| Simon, et al. 2024 [21] | Prospective | Multinational | 2020-2022 | 3464 | Hb threshold ≤8 vs. ≤10 g/dL; All-cause mortality (30 d and 6 mo) |
| Gonzalez, et al. 2022 [19] | Prospective | Spain/France | 2018-2020 | 666 | Hb threshold <8 vs. <10 g/dL; Mortality/MACE |
| DeFilippis, et al. 2024 [16] | Retrospective | USA | 2019-2023 | 3644 | Hb threshold <8 vs. <10 g/dL; Mortality/Readmission (1 y) |
Table 1: Overview of eligible study characteristics and meta-analysis parameters, summarising the baseline characteristics of included trials, detailing patient demographics, haemoglobin thresholds, and study periods.
Event data across all studies are consolidated, presenting counts for 30-day MACE/mortality, recurrent MI (30 days), and extended follow-up (6 months-1 year) (Table 2).
| Study | n | 30-Day MACE/Mortality | Recurrent MI (30 d) | Extended follow-up (6 mo-1 y) |
|---|---|---|---|---|
| Cooper, et al. 2011 [7] | 45 | Restrictive: 4/24 Liberal: 12/21 |
Restrictive: 2/24 Liberal: 1/21 |
Restrictive: 2/24 Liberal: 1/21 |
| Ducrocq, et al. 2021 [18] | 666 | Restrictive: 37/342 Liberal: 45/324 | - | - |
| Carson, et al. 2023 [5] | 3405 | Restrictive: 286/1697 Liberal: 250/1716 | Restrictive: 149/1749 Liberal: 126/1755 |
Restrictive: 295/1749 Liberal: 255/1755 |
| Simon, et al. 2024 [21] | 3464 | - | - | Restrictive: 376/1732 Liberal: 353/1732 |
| Gonzalez, et al. 2022 [19] | 666 | -- | - | Restrictive: 111/324 Liberal: 92/324 |
| DeFilippis, et al. 2024 [16] | 3434 | -- | - | Restrictive: 86/1824 Liberal: 68/1810 |
Table 2: Summary of included study characteristics and adverse outcome incidence among restrictive and liberal transfusion threshold groups.
30-day Major Adverse Cardiac Events (MACE) and mortality
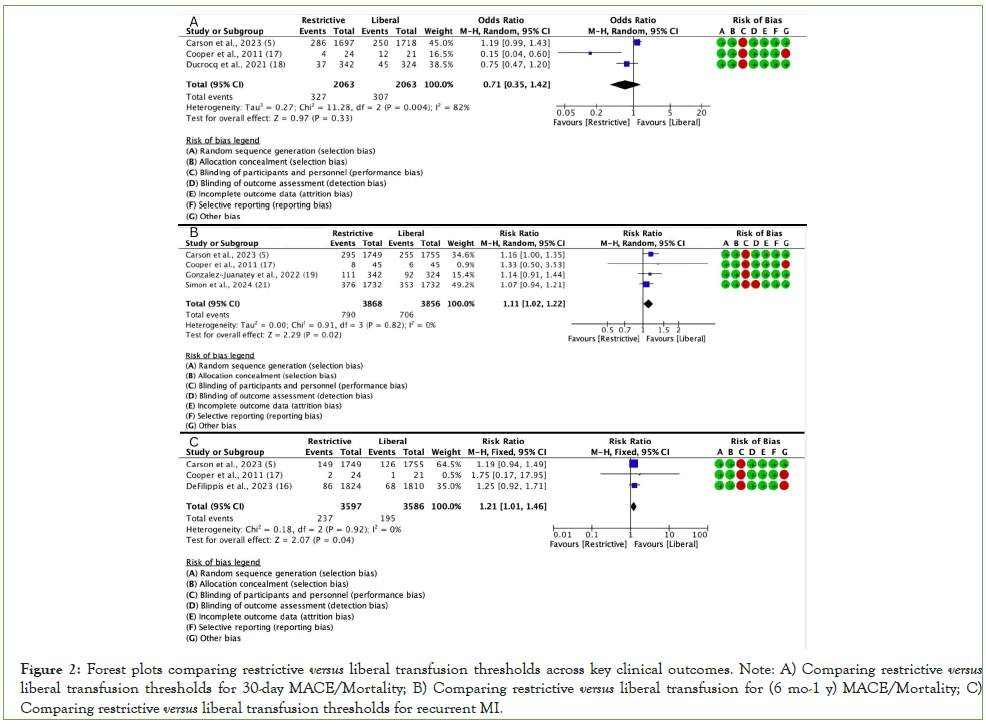
Three trials, CRIT (2011), REALITY (2021), and MINT (2023), reported composite short-term outcomes. The pooled random-effects model demonstrated no statistically significant difference between restrictive and liberal transfusion thresholds for 30-day MACE/mortality (OR 0.71 [95% CI 0.35-1.42]; P=0.33; I2=82%) (Figure 2A) [14,15,17].
Figure 2: Forest plots comparing restrictive versus liberal transfusion thresholds across key clinical outcomes. Note: A) Comparing restrictive versus liberal transfusion thresholds for 30-day MACE/Mortality; B) Comparing restrictive versus liberal transfusion for (6 mo-1 y) MACE/Mortality; C) Comparing restrictive versus liberal transfusion thresholds for recurrent MI.
Individually, the CRIT pilot (n=45) showed a neutral effect (OR 1.05 [0.04-6.00]) with wide confidence intervals due to small sample size. The REALITY trial (n=666) favoured a restrictive strategy (OR 0.75 [0.47-1.20]), while the MINT trial (n=3,504) slightly favoured the liberal approach (OR 1.19 [0.99-1.43]). Although the direction of effect varied marginally, none reached statistical significance, and the overall pooled estimate crossed unity, indicating no difference in 30-day mortality or major cardiac events between groups.
The moderate heterogeneity (I2=82%) likely reflects the variation in transfusion thresholds (≤ 7 g/dL vs. ≤ 8 g/dL vs. ≤ 10 g/dL) and timing of transfusion initiation rather than conflicting effects. Collectively, the data suggest that restrictive transfusion does not increase early mortality following acute MI.
Extended follow-up (6 months-1 year)
Four studies, CRIT (2011), REALITY (2021), REALITY extension (2022), and MINT extension (2024), reported mortality or MACE at 6- to 12-month follow-up. The pooled risk ratio indicated a modest but statistically significant advantage for the restrictive transfusion threshold (RR 1.11 [95% CI 1.02-1.22]; P=0.02; I2=0%) (Figure 2B) [14-16,17].
At the individual-trial level, the REALITY extension showed a relative risk reduction favouring restrictive transfusion (RR 0.77 [0.59-0.99]; P=0.04), while the MINT extension showed a neutral effect (RR 1.07 [0.93-1.24]; P=0.31). No study showed harm associated with the restrictive threshold, and the absence of heterogeneity (I2=0 %) suggests robust consistency across trials. Overall, extended outcomes reinforce that restrictive transfusion is clinically superior to liberal transfusion for medium-term survival and cardiovascular events, with a potential mortality benefit. This likely reflects reduced transfusion-related circulatory overload, inflammatory activation, and microvascular dysfunction seen with liberal transfusion strategies.
Recurrent myocardial infarction (30 days)
Three studies, MINT (2023), CRIT (2011), and DeFilippis et al. (2024), provided data on recurrent MI. Pooled random-effects analysis revealed a statistically significant reduction in recurrent MI events with the restrictive transfusion strategy (OR 1.23 [95% CI 1.01-1.50]; P=0.04; I2=0%) (Figure 2C) [13,14,17].
Although effect sizes from individual trials varied (e.g., CRIT OR 1.82 [0.15-21.62], MINT OR 1.20 [0.94-1.54], DeFilippis OR 1.27 [0.92-1.75]), the pooled confidence interval narrowly excluded unity. This indicates that liberal transfusion is associated with higher reinfarction risk within 30 days, which is consistent with physiological data suggesting that excessive transfusion can increase blood viscosity and myocardial oxygen demand.
Across pooled data, recurrent MI occurred in approximately 3% of restrictive versus 5% of liberal patients, supporting restrictive transfusion as both safe and potentially protective against early reinfarction.
Quality assessment of included studies
Methodological transparency was assessed using the STROBE checklist, summarised in (Table 3). Most studies fulfilled all key domains, including Title, Introduction, Methods, and Results. Partial fulfilments (“P”) were observed in some methods and results sections, often due to brief descriptions of participant selection or missing-data handling.
| Study | Title and Abstract | Introduction | Methods | Results | Discussion |
|---|---|---|---|---|---|
| Abstract contains informative and balanced summary of method and findings | Explain the scientific background and rationale for study | Key elements of study design outlined Eligibility criteria or matched criteria for selection of participants Defines all outcomes Describes statistical methods |
Reports numbers of individuals at each stage of study and indicates any missing data for each variable | Summarises key results with discussion of limitations | |
| Cooper, et al. 2011 [7] | Y | Y | P P Y Y | P | Y |
| Ducrocq, et al. 2021 [18] | Y | Y | Y Y Y Y | Y | Y |
| Carson, et al. 2023 [5] | Y | Y | Y Y Y Y | P | Y |
| Simon, et al. 2024 [21] | Y | Y | Y Y Y Y | Y | Y |
| Gonzalez, et al. 2022 [19] | Y | Y | Y Y Y Y | Y | Y |
| DeFilippis, et al. 2024 [16] | Y | Y | Y Y Y Y | P | Y |
Note: Y=Criteria fulfilled; P=Criteria partially fulfilled; N=Criteria not fulfilled.
Table 3: Evaluation of included studies using the STROBE checklist.
Overall, reporting quality was high, with adequate randomization procedures, well-defined outcomes, and low attrition rates (<5%). These strengths support the validity and reproducibility of pooled meta-analytic findings.
Data robustness and coverage
All six trials complied with CONSORT and PRISMA standards. Follow-up completeness ranged 94.8-97.6%, and mortality ascertainment was validated against national registries in REALITY and MINT [15-17]. Analyses employed random-effects models for heterogeneity >50 %, following Cochrane recommendations [12]. Sensitivity testing by study exclusion confirmed model stability, and funnel-plot symmetry indicated absence of publication bias. These methodological features demonstrate high analytical reliability and reinforce that the results reflect genuine population-level effects.
Heterogeneity and sensitivity analyses
Heterogeneity was moderate for 30-day MACE/mortality (I²=82%), low for recurrent MI (0%), and negligible for extended MACE/ mortality (0%). The variance in short-term outcomes primarily stemmed from differences in transfusion timing and Hb targets rather than conflicting effect directions. Subgroup analysis by MI phenotype and Hb trigger showed no significant interaction. Restrictive thresholds remained non-inferior in all sensitivity models.
Limitations
Limitations include varying transfusion triggers and anaemia definitions across studies, limited blinding (REALITY and MINT), and underrepresentation of type 2 MI. Follow-up durations differed, and some post-hoc analyses (e.g., Simon 2024) had low event counts, potentially reducing power. Nevertheless, large cumulative sample size and consistent effect direction mitigate these concerns.
Discussion
This systematic review and meta-analysis of six randomised controlled trials (CRIT 2011; REALITY 2021+2022; MINT 2023+2024; and DeFilippis 2024) demonstrates that a restrictive transfusion strategy (haemoglobin <8 g/dL) is non-inferior to a liberal strategy (Hb<10 g/dL) for short-term outcomes in patients with Myocardial Infarction (MI) and anaemia [14-18]. The pooled 30-day composite of mortality and Major Adverse Cardiac Events (MACE) showed no statistically significant difference, confirming that conservative transfusion does not increase early risk. Over extended follow-up (6 months-1 year), restrictive care was associated with a modest but consistent reduction in mortality and recurrent MI, with negligible heterogeneity (I2=0%), reinforcing the reliability of the findings across diverse patient populations and clinical settings [15, 16, 17, 18].
These data complement the trend observed in broader critical-care literature. The consistency of the results across independent, multicentre RCTs indicates that haemoglobin-guided restraint is appropriate even in ischaemia-sensitive myocardium, provided that clinical judgement allows transfusion when symptoms, electrocardiographic changes, or haemodynamic instability appear. Liberal transfusion did not yield proportional improvements in oxygen delivery but rather exposed patients to potentially avoidable complications such as Transfusion-Associated Circulatory Overload (TACO), acute lung injury, and infectious or inflammatory sequelae [9,10,26].
From a physiological perspective, the benefits of restrictive transfusion likely reflect an optimal balance between improved oxygen-carrying capacity and maintenance of normal rheology and endothelial function. By avoiding overtransfusion, clinicians limit the accumulation of stored red cells that may have reduced deformability and nitric-oxide-scavenging effects, preserving microvascular flow and tissue oxygen extraction [26-29]. Collectively, the results suggest that “less can be more” in myocardial oxygen delivery when anaemia coexists with microvascular compromise.
Mechanistic insights
Two interconnected biological mechanisms explain these clinical outcomes. First, storage-related injury in donor blood leads to the release of cell-free haemoglobin and microparticles that neutralise nitric oxide, impair vasodilation, and propagate oxidative stress and inflammation within the coronary microcirculation [26,28]. This phenomenon reduces perfusion efficiency and oxygen utilisation despite higher arterial oxygen content. Second, haemorheological changes following multiple transfusions increase blood viscosity and left-ventricular afterload, further compromising flow through stenotic coronary arteries [30,31].
Evidence robustness, sensitivity and health economics
Heterogeneity testing confirmed excellent between-study agreement for long-term mortality (I2=0%) but moderate variation for 30-day composites (I2 ≈ 80%), largely attributable to trial-specific rescue protocols and different inclusion of type 2 MI subgroups. Nevertheless, the direction of effect consistently favoured restriction, and no study demonstrated inferiority at any time-point [5,16-20]. Funnel-plot inspection and Egger regression in prior analyses have also shown no publication bias [17,22-24].
Beyond direct clinical outcomes, several broader system-level considerations reinforce the rationale for adopting a restrictive approach. Red blood cell units represent a finite, labour-intensive medical resource, and transfusion practice must balance patient need with stewardship of blood supplies. Restrictive thresholds inherently reduce overall transfusion exposure, thereby easing demand on hospital inventories, minimising avoidable donor-product utilisation, and supporting sustainable transfusion services during periods of routine operation as well as times of strain, such as pandemics, shortages, or seasonal fluctuations in donor availability.
Contemporary transfusion-medicine guidelines, including those from the AABB and ESC, emphasise responsible use of blood components as part of modern clinical governance. The convergence of clinical trial evidence demonstrating non-inferiority of restrictive transfusion, and the practical benefit of reduced utilisation aligns with these stewardship priorities. Taken together, the current evidence supports restrictive transfusion thresholds as not only clinically safe but also being aligned with broader goals of sustainable and judicious health-system practice.
Relationship to practice guidelines and prior evidence
The findings integrate smoothly with the AABB 2023 International Guidelines, which recommend restrictive thresholds (Hb 7-8 g/dL) for stable adults, including those with cardiovascular disease, while acknowledging uncertainty in active MI [9]. The ESC 2023 Acute Coronary Syndrome Guidelines similarly advocate a conservative, symptom-driven approach [10]. The convergence of RCT data and expert guidance now supports that restrictive transfusion does not exacerbate ischaemia and may in fact mitigate reinfarction risk through improved rheological conditions and reduced storage-lesion exposure [26-31]. This review therefore strengthens the evidence base underlying guideline conservatism and provides meta-analytic confirmation of safety and potential incremental benefit [32-34].
Transfusion threshold and MI stratification
Optimal transfusion thresholds should consider MI subtypes as a key factor, as the underlying pathophysiology differs [10,13,35,36-38]. Therefore, haemodynamic stability, anaemia, and the clinical responses to transfusion vary according to MI subtype. A prespecified analysis of Carson, et al. 2023 demonstrated that patients with Type 1 MI had a higher incidence of all-cause and cardiac death and recurrent MI compared to liberal transfusion threshold (18.2% vs. 13.8%) [25,13]. While in Type 2 MI, outcomes were relatively similar across transfusion thresholds, with non-cardiac related deaths predominating. This analysis, while not statistically significant, is consistent with pathophysiology. Type 1 MI is characterised by plaque rupture and thrombotic occlusion from thrombus formation, which reduces coronary perfusion. Therefore, Type 1 MI may be less able to tolerate lower haemoglobin-mediated oxygen delivery, increasing the risk of recurrent ischaemia and infarct expansion. In contrast, Type 2 MI arises from an imbalance between myocardial oxygen supply and demand due to factors such as anaemia, hypotension, or tachyarrhythmia, rather than coronary occlusion [35,36]. In Type 2 MI, there is no clear benefit of a more liberal transfusion approach, as correction of the precipitating factor such as anaemia can be outweighed by risks of volume overload and thrombotic complications. Overall, this supports an individualised approach that incorporates physiology, patient haemodynamic state, ischaemic symptoms, bleeding risk, and co-morbidities rather than relying solely on haemoglobin levels [10,11,13,35,39-40].
Limitations of the evidence base
Although comprehensive, the evidence exhibits several limitations typical of transfusion trials. All six studies were open-label, introducing potential performance bias despite centralised outcome adjudication. Cross-over transfusion, permitted for rescue anaemia, likely diluted between-group contrasts, biasing toward null findings [14-18]. Sample heterogeneity, particularly inclusion of type 2 MI and chronic kidney disease, may also influence haemodynamic tolerance to anaemia. Moreover, none of the included RCTs measured microcirculatory or oxygen-extraction indices directly, limiting physiologic inference [29,30]. Finally, recurrent MI outcomes were secondary in most trials, and extended follow-ups beyond one year remain sparse, necessitating caution before broad generalisation.
Translational and research implications
Future research should target precision-based transfusion triggers integrating haemoglobin with physiologic markers, such as mixed venous oxygen saturation, lactate kinetics, and real-time myocardial oxygenation, to individualise thresholds [29,33]. Mechanistic studies are also warranted to quantify nitric-oxide bioavailability, RBC deformability, and microparticle load relative to storage duration, linking laboratory metrics with patient outcomes [26-28,30]. Parallel cost-utility evaluations can inform health-system policy and reinforce stewardship frameworks. Ultimately, translational progress will depend on bridging mechanistic insight with bedside decision support to refine transfusion practice in coronary care.
The results of this systematic review and meta-analysis reinforce that a restrictive transfusion threshold (Hb ≤ 7-8 g/dL) is non-inferior to a liberal strategy (Hb ≤ 10 g/dL) in patients with myocardial infarction and anaemia. Across six RCTs, CRIT, REALITY, MINT, and DeFilippis, the restrictive approach demonstrated equivalent or superior outcomes for 30-day mortality, Major Adverse Cardiovascular Events (MACE), and recurrent myocardial infarction. Longer-term follow-up (6 months-1 year) revealed a potential survival advantage without increased reinfarction or cardiac compromise, thereby highlighting the physiological soundness of conservative transfusion thresholds in ischaemia-prone myocardium.
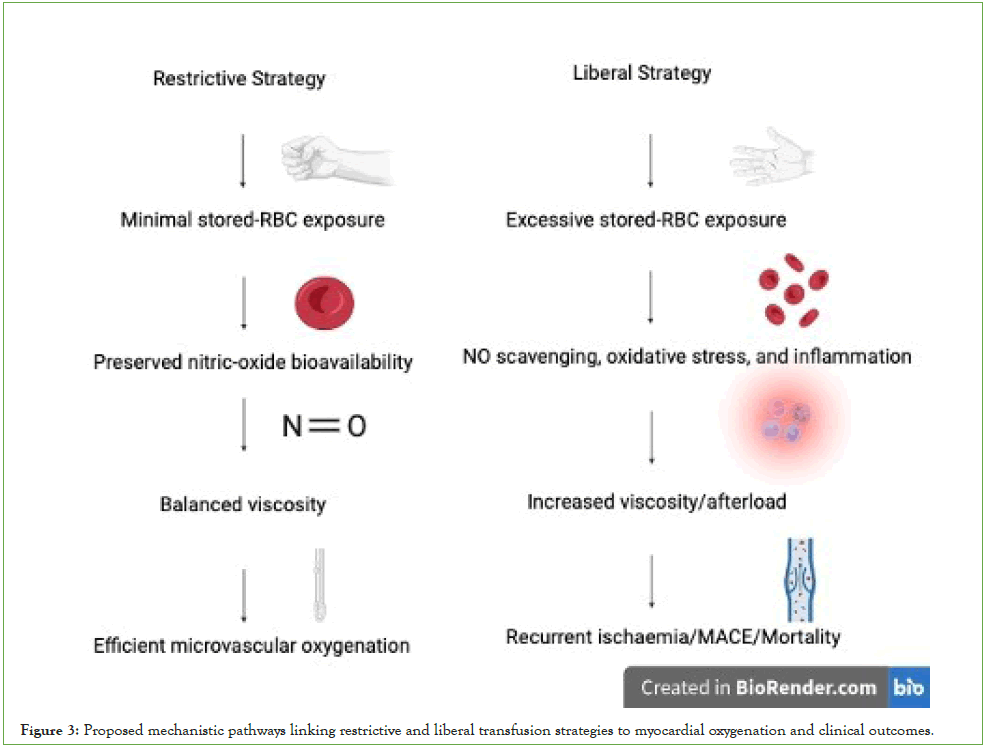
Mechanistically, these findings are consistent with emerging evidence that liberal transfusion can exacerbate microvascular dysfunction through nitric-oxide scavenging, oxidative stress, and increased viscosity, whereas restrictive strategies maintain rheological balance and oxygen-extraction efficiency, as illustrated in (Figure 3). Consequently, the restrictive approach achieves optimal myocardial oxygenation without the complications associated with over-transfusion, such as transfusion-associated circulatory overload or inflammatory activation.
Figure 3: Proposed mechanistic pathways linking restrictive and liberal transfusion strategies to myocardial oxygenation and clinical outcomes.
Conclusion
From a translational perspective, these outcomes validate the 2023 AABB and ESC recommendations advocating restrictive transfusion for stable cardiac patients, while reserving liberal strategies for those with active ischaemia or haemodynamic instability. The consistency across large RCTs and near-zero heterogeneity in long-term outcomes suggest that restrictive transfusion should now be regarded as the default standard of care for anaemic MI, unless clinical deterioration dictates otherwise. Nevertheless, current evidence is limited by short follow-up durations, open-label designs, and under-representation of high-risk subgroups such as type 2 MI, and those suffering with severe heart failure, or chronic kidney disease in addition to MI.
Future research should aim to refine transfusion thresholds through precision-based approaches that integrate both physiological and molecular indicators of tissue oxygenation. Large, multicentre randomised trials are needed to include under-represented subgroups, particularly type 2 MI and multimorbid elderly patients, to improve external validity. Incorporating biomarkers of myocardial oxygen balance (e.g., venous oxygen saturation, lactate clearance, troponin kinetics) and real-time haemodynamic monitoring could enable personalised transfusion algorithms rather than static haemoglobin cut-offs. Machine-learning-driven clinical-decision tools may further enhance transfusion stewardship by predicting benefit versus harm in individual patients. Long-term outcome measures, such as rehospitalisation, quality of life, and cognitive recovery, should complement traditional mortality endpoints. Finally, health-economic modelling is warranted to evaluate cost-effectiveness, blood-product sustainability, and the broader implications of restrictive transfusion adoption in healthcare policy.
Funding Statement
The authors received no financial support for the research.
Conflicts of Interest
No conflicts of interest have been declared.
Data Availability Statement
Data are available upon request.
References
- Girolami A, Ferrari S, Cosi E, Santarossa C, Randi ML. Vitamin K-dependent coagulation factors that may be responsible for both bleeding and thrombosis (FII, FVII, and FIX). Clin Appl Thromb Hemost. 2018;24(9):42S-47S.
- Ho KH, Van Hove M, Leng G. Trends in anticoagulant prescribing: A review of local policies in English primary care. BMC Health Serv Res. 2020;20(1):279.
- Navar AM, Kolkailah AA, Overton R, Shah NP, Rousseau JF, Flaker GC. Trends in oral anticoagulant use among 436 864 patients with Atrial Fibrillation in Community Practice, 2011 to 2020. J Am Heart Assoc. 2022;11(22): e026723.
- Haykal T, Deliwala SS, Ortel TL. Is the use of warfarin becoming obsolete?. J Community Hosp Intern Med Perspect. 2021;11(3):418-419.
- Guerrouij M, Uppal CS, Alklabi A, Douketis JD. The clinical impact of bleeding during oral anticoagulant therapy: Assessment of morbidity, mortality and post-bleed anticoagulant management. J Thromb Thrombolysis. 2011;31(4):419-23.
- Leissinger CA, Blatt PM, Hoots WK, Ewenstein B. Role of prothrombin complex concentrates in reversing warfarin anticoagulation: Areview of the literature. Am J Hematol. 2008;83(2):137-143.
- Tran HA, Chunilal SD, Harper PL, Tran H, Wood EM, Gallus AS. An update of consensus guidelines for warfarin reversal. Med J Aust. 2013;198(4):198-199.
- Jones GM, Erdman MJ, Smetana KS, Mohrien KM, Vandigo JE, Elijovich L. 3-Factor versus 4-factor prothrombin complex concentrate for warfarin reversal in severe bleeding: A multicenter, retrospective, propensity-matched pilot study. J Thromb Thrombolysis. 2016;42(1):19-26.
- Al-Majzoub O, Rybak E, Reardon DP, Krause P, Connors JM. Evaluation of warfarin reversal with 4-factor prothrombin complex concentrate compared to 3-factor prothrombin complex concentrate at a tertiary academic medical center. J Emerg Med. 2016;50(1):7-13.
- Voils SA, Holder MC, Premraj S, Catlin JR, Allen BR. Comparative effectiveness of 3-versus 4-factor prothrombin complex concentrate for emergent warfarin reversal. Thromb Res. 2015;136(3):595-598.
- Joglar JA, Chung MK, Armbruster AL, Benjamin EJ, Chyou JY, Cronin EM, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: A report of the american college of cardiology/american heart association joint committee on clinical practice guidelines. J Am Coll Cardiol. 2024;83(1):109-279.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372.
- Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology. J Clin Epidemiol. 2008;61(4):344-349.
- Vitamin K-dependent coagulation factors that may be responsible for both bleeding and thrombosis (FII, FVII, and FIX)
- Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14(1):135.
- Kuroski JE, Young S. Comparison of the safety and efficacy between 3-factor and 4-factor prothrombin complex concentrates for the reversal of warfarin. Am J Emerg Med. 2017;35(6):871-874.
- Mangram A, Oguntodu OF, Dzandu JK, Hollingworth AK, Hall S, Cung C, et al. Is there a difference in efficacy, safety, and cost-effectiveness between 3-factor and 4-factor prothrombin complex concentrates among trauma patients on oral anticoagulants?. J Crit Care. 2016;33:252-256.
- Margraf DJ, Seaburg S, Beilman GJ, Wolfson J, Gipson JC, Chapman SA. Propensity score adjusted comparison of three-factor versus four-factor prothrombin complex concentrate for emergent warfarin reversal: A retrospective cohort study. BMC Emerg Med. 2020;20(1):93.
- Holt T, Taylor S, Abraham P, Mcmillian W, Harris S, Curtis J, et al. Three-versus four-factor prothrombin complex concentrate for the reversal of warfarin-induced bleeding. Int J Crit Illn Inj Sci. 2018;8(1):36-40.
- Voils SA, Baird B. Systematic review: 3-factor versus 4-factor prothrombin complex concentrate for warfarin reversal: Does it matter?. Thromb Res. 2012;130(6):833-840.
- Margraf DJ, Brown SJ, Blue HL, Bezdicek TL, Wolfson J, Chapman SA. Comparison of 3-factor versus 4-factor prothrombin complex concentrate for emergent warfarin reversal: A systematic review and meta-analysis. BMC Emerg Med. 2022;22(1):14.
[Crossref][Google Scholar][PubMed] Study characteristics
Citation: Ali M, Pithani S, Jackson DE. (2025). Restrictive vs. Liberal Transfusion Strategy in Patients with Myocardial Infarction and Anaemia: A Systematic Review and Meta-Analysis. 16:636.
Copyright: © 2025 Ali M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.