Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2025) Volume 16, Issue 6
Protective and Therapeutic Effect of Blackberry Extract in Experimental Fibrosis Model
Seda Yilmaz1*, Metin Bagci1, Hasan Ibrahim Kozan2, Zuleyha Doganyigit3, Abdulkadir Basturk1, Seher Yilmaz4, Aslı Okan Oflamaz3 and Sumeyye Ucar52Meram Vocational School, University of Necmettin Erbakan, Konya, Turkey
3Department of Histology and Embryology, Faculty of Medicine, Yozgat Bozok University, Yozgat, Turkey
4Department of Anatomy, Faculty of Medicine, Yozgat Bozok University, Yozgat, Turkey
5Department of Anatomy, Faculty of Medicine, Erciyes University, Kayseri, Turkey
Received: 30-Jul-2025, Manuscript No. JBDT-25-29468; Editor assigned: 01-Aug-2025, Pre QC No. JBDT-25-29468 (PQ); Reviewed: 15-Aug-2025, QC No. JBDT-25-29468; Revised: 27-Aug-2025, Manuscript No. JBDT-25-29468 (R); Published: 29-Aug-2025, DOI: 10.4172/2155-9864.25.16.637
Abstract
Background: Bleomycin is a chemotherapeutic agent that can be used in many malignancies, but the side effect of pulmonary fibrosis plays a limiting role for treatment. Blackberry has been found to reduce oxidative stress, mainly because it is rich in bioactive components. The aim of this study was to evaluate whether blackberry extract could have a protective effect for the lung and a therapeutic effect in terms of fibrosis in bleomycin-induced experimental fibrosis model.
Methods: In the experimental study, 60 male sprague dawley rats weighing 180-200g were used.They were divided into 6 experimental groups. The doses of bleomycin, methylprednisolone and blackberry extract to be administered to the rats were selected in accordance with the literature, and the experiment was designed by administering these substances both before and after bleomycin for 14 days to determine whether blackberry extract has a protective or therapeutic effect. Histological and biochemical analysis of lung tissue samples were performed.
Results: Intra-alveolar haemorrhage was statistically significantly reduced in the bleomycin+blackberry group compared to the bleomycin group, and cellular infiltration and thickening of the alveolar wall were found to be less in the blackberry treated groups compared to the bleomycin group. The amount of collagen accumulated in bleomycin+blackberry group was less than bleomycin and blackberry+bleomycin groups. Malondialdehyde and total oxidant levels among oxidant parameters decreased and total antioxidant levels among antioxidant parameters increased in blackberry treated groups.
Conclusion: These results suggest that blackberry prevents bleomycin-induced pulmonary fibrosis in rats by reducing inflammatory and oxidant markers.
Keywords
Blackberry; Bleomycin; Fibrosis; Oxidative stress
Introduction
Bleomycin (BLE) is a glycopeptide antibiotic used as an antineoplastic agent and which has a wide range of applications including malignant conditions such as squamous cell carcinoma, lymphomas, testicular cancer, Kaposi sarcoma, osteosarcoma and even for pleurodesis [1-5].
The major limitation of BLE treatment is that life-threatening interstitial pulmonary fibrosis (also called fibrosing alveolitis) may develop in about 10% of patients receiving the treatment [6-9]. It causes reservations in terms of pulmonary complications especially in patients with a history of lung disease and smokers. The mechanism of BLE-induced lung injury is not completely clear. Oxidative damage in the lung appears to be important in the pathophysiology of lung injury, and antioxidants may improve the process [8-12].
Blackberry Extract (BBE) contains a wide range of active phytochemical components with anthocyanins and polyphenols. Therefore, its antioxidant and anti-inflammatory effects are prominent [13]. For this reason, this study was planned to answer the question whether the effects of BLE, which is a proinflammatory component, can be prevented by BBE, an antiinflammatory product.
Materials and Methods
The number of rats in the experimental groups was determined as the minimum number of 10 rats at the beginning of the group after the biostatistical power analysis. This study was carried out with the approval of Erciyes University Animal Experiments Local Ethics Committee (Protocol number: 23/058). All experimental procedures were carried out in accordance with ethical rules. In the experimental study, 60 male sprague dawley rats weighing 180-200 g were used and the rats were randomly divided into 6 experimental groups. During the experiment, rats were housed in cages with a constant ambient temperature (24 ± 2°C), humidity (60 ± 5%) and alternating 12 hours light and 12 hours dark cycles.
Chemical agents to be used in the experiment
Bleomycin: BLE, an important exogenous substance that triggers fibrosis in animal models, is prominent [14,15]. In a study in which BLE induced fibrosis in rats in the literature, 5 mg/kg was given in 180-200 g albino ann-marie male rats [16]. Based on the information presented, the Sprague dawley rat strain, which has an advantage in surgical procedures, was used in this project. In order to induce the experimental fibrosis model, BLE (Kocak, Istanbul, Turkey) was administered intratracheally at a dose of 5 mg/kg.
Blackberry Extract: BBE active metabolites have been shown to be protective against oxidative damage as well as showing strong antioxidant effect. Besides antioxidative effect, it also has antiinflammatory, antibacterial and anti-proliferative effects [17-19]. In a study, BBE was administered at different doses and a dosedependent decrease in oxidative activity was found [13]. In accordance with the information presented, BBE was administered to sprague dawley rats at a dose of 100 mg/kg gavage to investigate the parameters related to fibrosis treatment and protective effect in the project.
Berry pomace water extract was obtained using the method described by Turan and Simsek [20]. Firstly, berry fruits were washed and squeezed by hand using a fine mesh strainer. The pomaces were taken from the strainer and 50 g of the fruit’s pomaces were blended with 500 mL of distilled water. The mixture was homogenized at 900 rpm for 2 min using an Ultra-Turrax homogenizer (WiseTis HG–15D, Daihan Scientific Co., Seoul, Korea) and stirred for 1 h with a magnetic stirrer and filtered with Whatman 1. A rotary evaporator (Heidolph Rotary Evaporator, Laborota 4001, Germany) with vacuum pump (KNF Laboport Vacuum Pump, Germany) was then used to concentrate the filtrate. The obtained concentrated pomace water extracts were kept at 4 C until used.
The antioxidant activity, Total Phenolic Content (TPC), and Total Flavonoid Content (TFC) of blackberry extracts were evaluated. The free radical scavenging activities of extracts were determined using 1, 1-Diphenyl-2-picrylhydrazyl, Sigma-Aldrich (DPPH) according to [21]. The absorbance was recorded at 517 nm using a spectrophotometer (UV-160 A, UV-Visible recording spectrophotometer, Shimadzu, Tokyo, Japan). The results were expressed as a percentage of free radical scavenging activity (%). The total phenolic contents of extracts were determined using the Folin-Ciocalteu method as described by [22]. The absorption has measured at 750 nm against a reagent blank in a spectrophotometer. The results were given as mg Gallic Acid Equivalents (GAE)/100 mL. Total flavonoid contents were determined and all procedures were applied according to the method reported by [23]. The absorbance was measured at 510 nm. The results were reported as mg of catechin (standard) equivalents (mg CE/100 mL). DPPH radical scavenging activity was found to be 75.08% ± 2.47, the TPC was quantified at 57.21 mg GAE/100 mL ± 0.80, and the TFC was measured to be 4.71 mg CE/100 mL ± 0.04. These values are presented as mean ± standard error.
Experimental groups
Control group (n=10): Rats in the control group were fed rodent rearing pellets with free access to feed and water for 14 days. Only Saline (SF) (0.9% NaCl sol.) 1 mL was administered intraperitoneally (i.p.) every day for 28 days.
Fibrosis group (n=10): BLE 0.5 mL at a dose of 5 mg/kg was administered intratracheally as a single dose [16].
Positive group (n=10): It will be used as a positive control. BLE 0.5 mL single dose of 5 mg/kg dose was administered intratracheally, and methylprednisolone at a dose of 4 mg/kg i.p. for 15 days after BLE administration [24].
BBE group (BBE) (n=10): BBE agent at a dose of 100 mg/kg was administered by 1 mL oral gavage every day for 28 days [13].
BBE+BLE group (n=10): BBE at a dose of 100 mg/kg was administered by oral gavage every day for 28 days. On the 15th day, a single dose of 5 mg/kg intratracheal BLE was administered [25].
BLE+BBE group (n=10): After 5 mg/kg single dose intratracheal BLE was administered, BBE was administered via oral gavage at a dose of 100 mg/kg for 28 days [25].
Histological analysis
Lung tissues obtained from the experimental groups were placed in 10% formaldehyde solution and fixed for 1 day. Following fixation, the tissues were placed under running water overnight. The next day, they were kept in increasing alcohol series (70%, 80% and 96%) for one day. After being left in 100% alcohol for 3 hours, xylol transparency was applied. They were left in paraffin I, II and III at one-hour intervals. The samples were then embedded in clean paraffin and paraffin blocks were made. From each paraffin block, 5 μm thickness sections were taken on slides. Sections were passed through xylol and decreasing alcohol series. Sections stained with haem atoxylin and eosin (H and E, BES LAB). The parameters of haemorrhage, cellular infiltration and alveolar wall thickening in the lung tissue were scored as 0 none, 1 mild, 2 moderate and 3 severe [26]. Lung tissue sections were stained using Masson's trichrome (Bio-Optica) technique to examine the histological features of pulmonary fibrosis. Images of Masson's trichrome staining of lung tissue sections was analysed quantitatively using Image J software 1.54f [27,28]. In images with Masson's trichrome staining, the blue colouration indicating extracellular matrix localisation was separated and measured as intensity per field of view.
Biochemical analyses
Malondialdehyde (MDA) (Sunredbio Catalog number, 201-11-0157, China), Total Antioxidant Status (TAS) (Sunredbio Catalog number, 201-11-2672, China) and Total Oxidant Status (TOS) (Sunredbio Catalog number, 201-11-1669, China) analyses in tissue samples of the experimental groups were performed according to the commercially available kit protocol. The results were read at 450 nm wavelength and expressed as nmol/mL for MDA and TOS and U/mL for TAS.
Statistical analysis
Numerical values of the scored histopathological findings and quantitative values of Masson trichrome staining intensity were compared by one-way analysis of variance using GraphPad Prism 8 Version 8.4.3 software. The difference between the groups was determined by applying Tukey's multiple comparison test. p<0.05 was considered statistically significant.
For biochemical analyses, Shapiro-Wilk test was applied to evaluate the normality and lognormality of the data. For normally distributed data, one-way ANOVA was used for multiple comparisons between groups. Kruskal-Wallis test was used for multiple comparisons between groups for non-normally distributed data.
Results
Histological analysis results
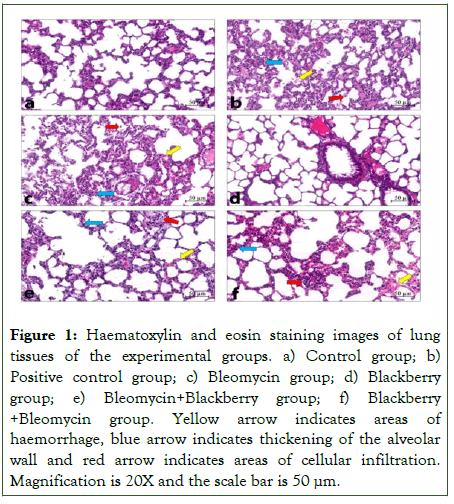
When the lung specimens stained with haematoxylin eosin were examined, it was observed that bronchioles, alveoli and pulmonary arteries had normal histology in control and BBE groups (Figure 1). Intraalveolar haemorrhage was observed to decrease in the BLE+BBE group compared to the BLE group. There was no significant difference in cellular infiltration and alveolar wall thickening in the BLE group compared to the BLE group (Table 1).
| Groups | Haemorrhage | Cellular infiltration | Alveolar wall thickening |
|---|---|---|---|
| Control group | 0.18 ± 0.07 | 0.15 ± 0.10 | 0.25 ± 0.05 |
| Positive control group | 1.05 ± 0.37a | 1.16 ± 0.16a | 1.5 ± 0.26a |
| Bleomycin group | 1.5 ± 0.37a | 1.61 ± 0.27ab | 1.56 ± 0.25a |
| Blackberry group | 0.2 ± 0.14bc | 0.18 ± 0.07bc | 0.28 ± 0.13bc |
| Bleomycin+Blackberry group | 0.88 ± 0.31acd | 1.41 ± 0.23ad | 1.18 ± 0.32ad |
| Blackberry+Bleomycin group | 1.61 ± 0.22abde | 1.46 ± 0.18ad | 1.21 ± 0.24ad |
Note: a: p<0.05 shows that there is a difference compared to control group; b: p<0.05 shows that there is a difference compared to positive control group; c: p<0.05 shows that there is a difference compared to bleomycin group; d: p<0.05 shows that there is a difference compared to Blackberry group; e: p<0.05 shows that there is a difference compared to Bleomycin+Blackberry group; f: p<0,05 shows that there is a difference compared to Blackberry+Bleomycin group.
Table 1: Damage rates observed in the lung tissues of the experimental groups.
Figure 1: Haematoxylin and eosin staining images of lung tissues of the experimental groups. a) Control group; b)Positive control group; c) Bleomycin group; d) Blackberrygroup; e) Bleomycin+Blackberry group; f) Blackberry +Bleomycin group. Yellow arrow indicates areas of haemorrhage, blue arrow indicates thickening of the alveolar wall and red arrow indicates areas of cellular infiltration. Magnification is 20X and the scale bar is 50 μm..
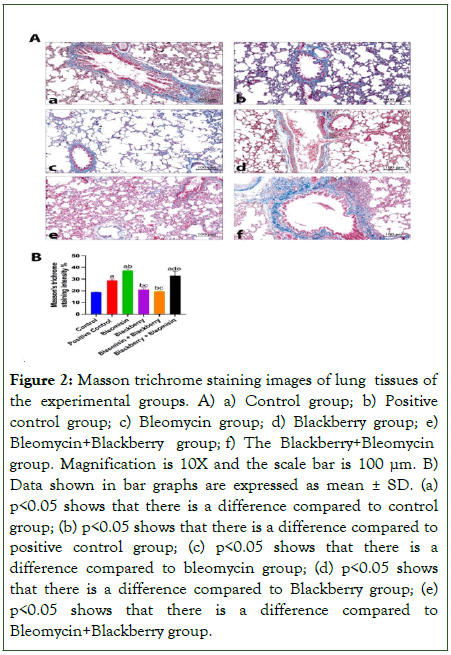
Images with Masson's trichrome staining was separated and quantified in Image J software, with blue colouration indicating collagen localisation as intensity per field of view. When these quantitative values were statistically tested, it was observed that collagen deposition increased significantly in the positive control, BLE and BBE+BLE groups compared to the control group (Figure 2, p<0.05). The amount of collagen deposited in the BLE+BBE group was less than the BLE and BBE+BLE groups (Figure 2, p<0.05).
Figure 2: Masson trichrome staining images of lung tissues of the experimental groups. A) a) Control group; b) Positive control group; c) Bleomycin group; d) Blackberry group; e) Bleomycin+Blackberry group; f) The Blackberry+Bleomycin group. Magnification is 10X and the scale bar is 100 μm. B) Data shown in bar graphs are expressed as mean ± SD. (a) p<0.05 shows that there is a difference compared to control group; (b) p<0.05 shows that there is a difference compared to positive control group; (c) p<0.05 shows that there is a difference compared to bleomycin group; (d) p<0.05 shows that there is a difference compared to Blackberry group; (e) p<0.05 shows that there is a difference compared to Bleomycin+Blackberry group.
Biochemical analysis results
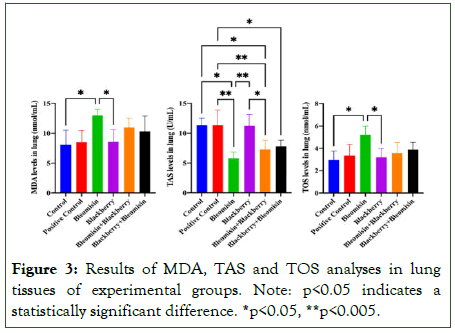
The results of biochemical analyses of lung tissues taken from the experimental groups are shown in (Figure 3). According to the data obtained, BLE application increases MDA and TOS levels and decreases TAS levels in a statistically significant manner compared to the control group. It is observed that BBE alleviates these negative effects.
Figure 3: Results of MDA, TAS and TOS analyses in lung tissues of experimental groups. Note: p<0.05 indicates a statistically significant difference. *p<0.05, **p<0.005.
Shapiro-Wilk test was applied to evaluate the normality and lognormality of the data. For normally distributed data, one-way ANOVA was used for multiple comparisons between groups. Kruskal-Wallis test was used for multiple comparisons between groups for non-normally distributed data.
Discussion
BLE is an antitumor antibiotic used in the treatment of various malignancies, including germ cell tumours and Hodgkin lymphoma. The most important and serious side effect is pulmonary toxicity. This adverse effect can lead to pulmonary fibrosis, sometimes chronic and irreversible, which limits its application, has been associated with an acquired loss of BLE hydrolase activity and is mediated by an immunological mechanism characterised by migration of active effector cells in the lung and release of proinflammatory mediators, resulting in the development of pulmonary fibrosis [29-33]. Athymic rats are resistant to BLE-induced lung injury, suggesting that the inflammatory process is important for the pathogenesis of the disease [34].
The importance of inflammatory cytokines in the development of BLE-induced lung injury has been suggested by various observations. Activation of messenger RNA-encoding cytokines after BLE exposure in whole lung and bronchoalveolar lavage preparations precedes the proliferative response and induction of collagen synthesis [28,35,36].
In animal models, neutralisation of the biological activity of proinflammatory cytokines by neutralising antibodies (e.g. anti-TNFα and anti-TGF-beta), soluble receptors (e.g. recombinant human TNFα receptors) or receptor antagonists that bind and inactivate the relevant peptides (e.g. interleukin-1 receptor antagonists) leads to amelioration of the lung fibrotic process induced by BLE [31,37,38].
The alveolar macrophage is considered to play a central role in the development of BLE-induced lung injury due to its ability to express a range of effector molecules (e.g. cytokines, lipid metabolites, oxygen radicals). The mechanism of activation of alveolar macrophages by BLE is unknown. BLE receptors have been identified on the surface of rat alveolar macrophages, suggesting that activation may be mediated by a second messenger [39].
In another study, BLE treatment was shown to induce fibroblast migration from the bone marrow to the lung [40]. These fibroblasts express type I collagen, telomerase and CXCR4 and CCR7 chemokine receptors.
Studies on antioxidant anti-inflammatory products that prevent and treat lung toxicity due to BLE are being carried out. Various herbal extracts have been used in studies on this subject and successful results have been obtained. The main aim of our study was to determine the success of BBE, which has a high antioxidant capacity, in preventing and treating BLE-induced lung toxicity [13].
BBE contains a wide range of active phytochemical components with anthocyanins and polyphenols. Therefore, its antioxidant and anti-inflammatory effect is prominent [13]. For this reason, this study was planned to answer the question whether the effects of BLE, a proinflammatory component, can be prevented by BBE, an anti-inflammatory product.
BBE has antibacterial and anti-proliferative effects as well as antioxidative and anti-inflammatory effects [17-19]. In our study, a statistically significant decrease in intra-alveolar haemorrhage was observed in the BLE+BBE group compared to the BLE group. Cellular infiltration and alveolar wall thickening were found to be proportionally decreased in the BLE groups treated with BBE compared to the BLE group, but there was no statistically significant difference between the groups. This difference may become significant in a study with larger experimental groups. In terms of collagen accumulation, the amount of collagen accumulated in the BLE+BBE group was found to be less than the BLE and BBE+BLE groups. These results of our study support that BBE has anti-inflammatory, antiproliferative effect.
In a study using kunming male rats weighing 18-22 g, BBE was administered intragastrically at 25, 50 and 100 mg/kg and a dose-dependent decrease in superoxide dismutase activity was found [13]. In our study, it was found that MDA and TOS decreased among oxidant parameters and TAS increased among antioxidant parameters in BLE-treated groups. BBE was found to be antioxidative and was found to alleviate the negative effects of BLE.
Conclusion
Bleomycin-related side effects may be prevented or treated with blackberry extract. We think that these results will prevent the reservations about bleomycin and will make an important contribution to the literature.
Funding Statements
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
SY, MB, HIK, AB contributed to conceptualization, methodology, collected and analyzed the literature; ZD, SY, AOO, SU contributed to formal analysis, the experiment contributed to the follow-up of the strainer and histopathological evaluations; SY writingreview and editing; SY and ZD conceived and gave the final approval of the submitted version. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
The raw data is available upon request.
Ethics Approval
The experimental protocol of this study was approved by Kayseri Erciyes University Animal Experiments Local Ethics Committee (Protocol number: 23/058).
Conflicts of Interest
The authors declare that they have no conflicts of interest to report regarding the present study.
References
- Meadors M, Floyd J, Perry MC. Pulmonary toxicity of chemotherapy. Semin Oncol. 2006;33(1):98-105.
- Lavoie JM, Kollmannsberger CK. Current management of disseminated germ cell tumors. Urol Clin North Am. 2019;46(3):377-388.
- Bennett JM, Reich SD. Drugs five years later: Bleomycin. Ann Intern Med. 1979;90(6):945-948.
- Kong J, Yi L, Xiong Y, Huang Y, Yang D, Yan X, et al. The discovery and development of microbial bleomycin analogues. Appl Microbiol Biotechnol. 2018;102(16):6791-6798.
- Bagheri R, Noori M, Rajayi M, Attaran D, Ashari AMHA, Lari SM, et al. The effect of iodopovidone versus bleomycin in chemical pleurodesis. Asian Cardiovasc Thorac Ann. 2018;26(5):382-386.
- Jules-Elysee K, White DA. Bleomycin-induced pulmonary toxicity. Clin Chest Med. 1990;11(1):1-20.
- Camus P. Interstitial lung disease from drugs, biologics, and radiation. Interstitial lung disease. 2011:637.
- Sleijfer S. Bleomycin-induced pneumonitis. Chest. 2001;120(2):617-624.
- O’sullivan JM, Huddart RA, Norman AR, Nicholls J, Dearnaley DP, Horwich A. Predicting the risk of bleomycin lung toxicity in patients with germ-cell tumours. Ann Oncol. 2003;14(1):91-96.
- Sikic BI. Biochemical and cellular determinants of bleomycin cytotoxicity. Cancer Surv. 1986;5(1):81-91. [Crossref]
- Chandler DB. Possible mechanisms of bleomycin-induced fibrosis. Clin Chest Med. 1990;11(1):21-30.
- Fantone JC, Phan SH. Oxygen metabolite detoxifying enzyme levels in bleomycin-induced fibrotic lungs. Free Radic Biol Med. 1988;4(6):399-402.
- Xiao T, Guo Z, Fu M, Huang J, Wang X, Zhao Y, et al. Amelioration of alcoholic liver disease by activating PXR-Cytochrome P450s axis with blackberry extract. Separations. 2022;9(10):321.
- Della Latta V, Cecchettini A, Del Ry S, Morales MA. Bleomycin in the setting of lung fibrosis induction: From biological mechanisms to counteractions. Pharmacol Res. 2015;97:122-130.
- Liu T, De Los Santos FG, Phan SH. The bleomycin model of pulmonary fibrosis. Methods Mol Biol. 2017;27-42.
- Samareh Fekri M, Poursalehi HR, Sharififar F, Mandegary A, Rostamzadeh F, Mahmoodi R. The effects of methanolic extract of Glycyrrhiza glabra on the prevention and treatment of bleomycin-induced pulmonary fibrosis in rat: Experimental study. Drug Chem Toxicol. 2021;44(4):365-371.
- Abeywickrama G, Debnath SC, Ambigaipalan P, Shahidi F. Phenolics of selected cranberry genotypes (Vaccinium macrocarpon Ait.) and their antioxidant efficacy. J Agric Food Chem. 2016;64(49):9342-9351.
- Kaneria M, Baravalia Y, Vaghasiya Y, Chanda S. Determination of antibacterial and antioxidant potential of some medicinal plants from Saurashtra region, India. Indian J Pharm Sci. 2009;71(4):406.
- Aqil F, Gupta A, Munagala R, Jeyabalan J, Kausar H, Sharma RJ, et al. Antioxidant and antiproliferative activities of anthocyanin/ellagitannin-enriched extracts from Syzygium cumini L.(Jamun, the Indian Blackberry). Nutr Cancer. 2012;64(3):428-438.
- Turan E, Å?imÅ?ek A. Effects of lyophilized black mulberry water extract on lipid oxidation, metmyoglobin formation, color stability, microbial quality and sensory properties of beef patties stored under aerobic and vacuum packaging conditions. Meat Sci. 2021;178:108522.
- Sharma OP, Bhat TK. DPPH antioxidant assay revisited. Food Chem. 2009;113(4):1202-1205.
- Singleton VL, Orthofer R, Lamuela-Raventós RM. [14] Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999;299:152-178.
[Crossref][Google Scholar][PubMed]
- Park YS, Jung ST, Kang SG, Heo BG, Arancibia-Avila P, Toledo F, et al. Antioxidants and proteins in ethylene-treated kiwifruits. Food Chem. 2008;107(2):640-648.
- Aytemur ZA, Hacievliyagil SS, Iraz M, Samdanci EM, Ozerol EL, Kuku I, et al. Effects of iloprost on bleomycin-induced pulmonary fibrosis in rats compared with methyl-prednisolone. Rev Port Pneumol. 2012;18(6):272-277.
- Moeller A, Ask K, Warburton D, Gauldie J, Kolb M. The bleomycin animal model: A useful tool to investigate treatment options for idiopathic pulmonary fibrosis? Int J Biochem Cell Biol. 2008;40(3):362-382.
- Passmore MR, Byrne L, Obonyo NG, See Hoe LE, Boon AC, Diab SD, et al. Inflammation and lung injury in an ovine model of fluid resuscitated endotoxemic shock. Respir Res. 2018;19(1):231.
- Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9(7):671-675.
- Togami K, Ozaki H, Yumita Y, Kitayama A, Tada H, Chono S. threeâ?dimensional imaging of pulmonary fibrotic foci at the alveolar scale using tissueâ?clearing treatment with staining techniques of extracellular matrix. Int J Biomed Imaging. 2020;2020(1):8815231.
- Filderman AE, Genovese LA, Lazo JS. Alterations in pulmonary protective enzymes following systemic bleomycin treatment in mice. Biochem Pharmacol. 1988;37(6):1111-1116.
- Phan SH, Kunkel SL. Lung cytokine production in bleomycin-induced pulmonary fibrosis. Exp Lung Res. 1992;18(1):29-43.
- Piguet PF, Collart MA, Grau GE, Kapanci YU, Vassalli PI. Tumor necrosis factor/cachectin plays a key role in bleomycin-induced pneumopathy and fibrosis. J Exp Med. 1989;170(3):655-663.
- Ortiz LA, Lasky J, Hamilton RF, Holian A, Hoyle GW, Banks W, et al. Expression of TNF and the necessity of TNF receptors in bleomycin-induced lung injury in mice. Exp Lung Res. 1998;24(6):721-743.
- Ballinger MN, Newstead MW, Zeng X, Bhan U, Mo XM, Kunkel SL, et al. IRAK-M promotes alternative macrophage activation and fibroproliferation in bleomycin-induced lung injury. J Immunol. 2015;194(4):1894-1904.
- Schrier DJ, Phan SH, McGarry BM. The effects of the nude (nu/nu) mutation on bleomycin-induced pulmonary fibrosis: A biochemical evaluation. Am Rev Respir Dis. 1983;127(5):614-617.
- Hoyt DG, Lazo JS. Alterations in pulmonary mRNA encoding procollagens, fibronectin and transforming growth factor-beta precede bleomycin-induced pulmonary fibrosis in mice. J Pharmacol Exp Ther. 1988;246(2):765-771.
- Khalil N, Whitman C, Zuo L, Danielpour D, Greenberg A. Regulation of alveolar macrophage transforming growth factor-beta secretion by corticosteroids in bleomycin-induced pulmonary inflammation in the rat. J Clin Invest. 1993;92(4):1812-1818.
- Giri SN, Hyde DM, Hollinger MA. Effect of antibody to transforming growth factor beta on bleomycin induced accumulation of lung collagen in mice. Thorax. 1993;48(10):959-966.
- Piguet PF, Vesin C, Grau GE, Thompson RC. Interleukin 1 receptor antagonist (IL-1ra) prevents or cures pulmonary fibrosis elicited in mice by bleomycin or silica. Cytokine. 1993;5(1):57-61.
- Denholm EM, Phan SH. Bleomycin binding sites on alveolar macrophages. J Leukoc Biol. 1990;48(6):519-523.
- Hashimoto N, Jin H, Liu T, Chensue SW, Phan SH. Bone marrow-derived progenitor cells in pulmonary fibrosis. J Clin Invest. 2004;113(2):243-252.
Citation: Yilmaz S, Bagci M, Kozan HI, Doganyigit Z, Basturk A, Yilmaz S, et al. (2025). Protective and Therapeutic Effect of Blackberry Extract in Experimental Fibrosis Model. J Blood Disord Transfus. 16:637.
Copyright: © 2025 Yilmaz S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.