Indexed In
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- CiteFactor
- Ulrich's Periodicals Directory
- Access to Global Online Research in Agriculture (AGORA)
- Electronic Journals Library
- Centre for Agriculture and Biosciences International (CABI)
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Scholarsteer
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Perspective - (2025) Volume 16, Issue 3
New Progress in the Phylogeny and Classification of the Largest Fern Family Dryopteridaceae
Li-Bing Zhang*Received: 13-Sep-2024, Manuscript No. JPPM-24-26925; Editor assigned: 16-Sep-2024, Pre QC No. JPPM-24-26925 (PQ); Reviewed: 30-Sep-2024, QC No. JPPM-24-26925; Revised: 22-Jun-2025, Manuscript No. JPPM-24-26925 (R); Published: 29-Jun-2025, DOI: 10.35248/2157-7471.25.16.762
Abstract
Dryopteridaceae are the largest fern family containing over 2,100 species worldwide found in various habitats. Previous studies identified three major clades and two subclades and proposed a classification with three subfamilies. However, some critical relationships remained unresolved and some genera remained unsampled. In a most recent study, all the genera of Dryopteridaceae were sampled. Complete chloroplast genomes for 91 species were obtained and a highly supported phylogenetic tree for all extant genera within the family was reconstructed. Based on the new phylogeny and morphological analysis, a classification with seven subfamilies and 24 genera was proposed.
Keywords
Ctenitoideae; Fern phylogenomics; Plastomes; Lastreopsidoideae; Pleocnemioideae; Polystichopsidoideae; Pseudarachniodes
Introduction
Current status
Ferns are the most diverse and abundant group among extant vascular plants, apart from angiosperms, with species numbering approximately five times that of gymnosperms and lycophytes combined. Although angiosperms dominate global ecosystems, ferns remain an important component of terrestrial plant ecosystems. With the rise of angiosperms during the Cretaceous period, other terrestrial plants gradually declined, but ferns evolved to adapt to various environments, resulting in morphological and species diversity within forests dominated by angiosperms. In the widely accepted classification for ferns, 48 families, 319 genera, and approximately 10,578 species of ferns (monilophytes, ferns) are recognized, with the core group Polypodiales (polypods) comprising over 250 genera and about 8,700 species [1].
Description
The family Dryopteridaceae, under the order Polypodiales, is the largest fern family, including over 2,100 species and some largest fern genera, e.g., Dryopteris adans, Polystichum Roth, accounting for one-fifth of all existing fern species. This family has a nearglobal distribution, with the highest diversity in temperate and tropical mountain regions. Plants of this family can be found in various habitats, from coastal areas to the alpine tree line. Most species in the family are terrestrial, while others are lithophytes, epiphytes, semi-epiphytes, or tropical climbing plants. The family exhibits a complex and diverse morphology (Figure 1), encompassing a wide range of forms seen within Polypodiales. Rhizomes can be creeping, erect, ascending, or occasionally climbing. The leaves can be monomorphic (not differentiated into fertile and sterile leaves, with both photosynthesis and spore production occurring on the same leaves) or dimorphic (with fertile and sterile leaves differing in shape), ranging from simple leaves to one to five times pinnately compound leaves. The veins can be pinnately arranged or reticulate, with sori usually clustered into round structures, occasionally scattered on the leaf underside, with or without an indusium [2-4].
Figure 1: Photographs of representative Dryopteridaceae taxa. A) Elaphoglossum yoshinagae (Yatabe) Makino; B) Bolbitis subcordata (Copel.) Ching; C) Olfersia cervina (L.) Kunze; D) Lomagramma sorbifolia (Willd.) Ching; E) Polybotrya altescandens C. Chr.; F) Stigmatopteris heterophlebia (Baker) R.C. Moran; G) Cyrtomium fortunei J. Sm.; H) Dryopteris wallichiana (Spreng.) Hyl.; I) Ctenitis decurrenti-pinnata (Ching) Tardieu and C. Chr.; J) Lastreopsis subrecedens Ching; K) Trichoneuron microlepioides Ching; L) Pleocnemia winitii Holttum.
Due to the worldwide distribution of the family, the uneven number of species across genera, and the complex morphological diversity, reconstructing the phylogeny of the family presents a significant challenge. He conducted a phylogenetic study of the family based on a comprehensive sampling, identifying three major clades and two subclades, and proposed a classification with three subfamilies (Dryopteridoideae, Elaphoglossoideae, and Polybotryoideae). However, several issues remained unresolved. First, the phylogenetic relationships among the three subfamilies were still not fully determined. Second, the systematic positions of the genera Ctenitis and Stigmatopteris remained unresolved and were temporarily placed in Dryopteridoideae and Polybotryoideae, respectively. Additionally, several smaller genera were not sampled in their study [5]. The PPG I classification accepted Liu et al.'s treatment, dividing the family into three subfamilies and 26 genera, with two genera of uncertain placement later proven to belong to the families Tectariaceae and Lomariopsidaceae.
New progress
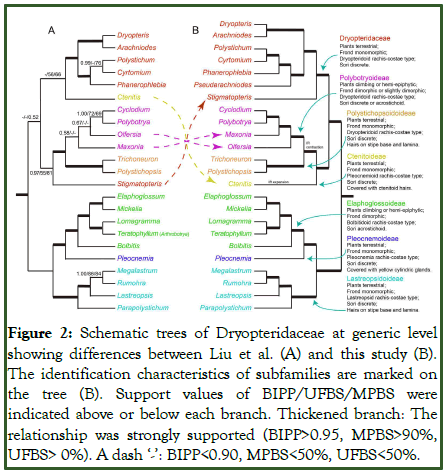
In the most recent study (Zuo et al., in press) all the genera of Dryopteridaceae recognized by PPG I were sampled. Complete chloroplast genomes for 91 species of Dryopteridaceae were obtained and a highly supported phylogenetic tree for all extant genera within the family was reconstructed. The phylogenetic analysis revealed seven major clades within Dryopteridaceae, further subdivided into 24 clades (Figure 2).
Figure 2: Schematic trees of Dryopteridaceae at generic level showing differences between Liu et al. (A) and this study (B). The identification characteristics of subfamilies are marked on the tree (B). Support values of BIPP/UFBS/MPBS were indicated above or below each branch. Thickened branch: The relationship was strongly supported (BIPP>0.95, MPBS>90%, UFBS> 0%). A dash ‘-’: BIPP<0.90, MPBS<50%, UFBS<50%.
To identify the morphological characteristics of each major clade and integrate broad morphological and molecular data, 13 morphological traits were selected for detailed analysis. The results showed that seven of these traits could be used to distinguish the seven major clades and 24 subclades within Dryopteridaceae. These traits include growth habit, rhizome morphology, leaf morphology, rachis-costal structure, appendages on the petiole base and leaf surface (such as scales, hairs, and bristles), and spore arrangement. Based on the phylogeny and morphological-trait analysis, the classification of Dryopteridaceae was updated and seven subfamilies and 24 genera were recognized, including four newly established subfamilies: Ctenitoideae, Lastreopsidoideae, Pleocnemioideae, and Polystichopsidoideae. In this classification, most genera previously recognized in PPG I are retained [6]. The genus Stigmatopetris, whose phylogenetic position was unresolved in earlier studies, is now placed in Dryopteridoideae, while Arthrobotrya is treated as a synonym of Teratophyllum. Additionally, a new endemic genus from Madagascar, Pseudarachniodes, was discovered and described [7].
The study, titled "A revised classification of Dryopteridaceae based on plastome phylogenomics and morphological evidence, with the description of a new genus, Pseudarachniodes," was published online in Plant Diversity (Zuo et al., in press). The robust phylogenetic framework and reconstructed morphological trait evolution offer new insights into the phylogenetic relationships among different lineages of Dryopteridaceae and provide a foundational framework for future research on the taxonomy, biogeography, and diversity of the family [8,9].
Conclusion
Currently, pteridologists are preparing to update the PPG classification, and we need to consider how to make the classification of ferns more practical. For example, when identifying species from large families such as Dryopteridaceae, Polypodiaceae, and Pteridaceae, each with over 1,000 species, how can we first determine the higher classification unit (family or subfamily) to which a species belongs, and then assign it to a lower classification unit (genus) based on morphological characteristics, rather than determining the genus first and then assigning it to the corresponding family? Clearly, monophyletic groups supported by both molecular and morphological evidence are more suitable for defining "natural" families or subfamilies.
References
- Du XY, Lu JM, Zhang LB, Wen J, Kuo LY, Mynssen CM, et al. Simultaneous diversification of Polypodiales and angiosperms in the Mesozoic. Cladistics. 2021;37:518–539.
[Crossref] [Google Scholar] [PubMed]
- Liu HM, Zhang XC, Wang MP, Shang H, Zhou SL, Yan YH, et al. Phylogenetic placement of the enigmatic fern genus Trichoneuron informs on the infra-familial relationship of Dryopteridaceae. Plant Syst Evol. 2016;302:319–332.
- PPG I. A community-derived classification for extant lycopods and ferns. J Syst Evol. 2016;54:563–603.
- Schneider H, Schuettpelz E, Pryer KM, Cranfill R, Magallón S, Lupia R. Ferns diversified in the shadow of angiosperms. Nature. 2004;428:553–557.
[Crossref] [Google Scholar] [PubMed]
- Zhang LB, Wu SG, Xiang JY, Xing FW, He H, Wang FG, et al. Dryopteridaceae. In: Wu ZY, Raven PH, Hong DY, eds. Flora of China, Vol. 2–3. 2013:542–724.
- Zuo ZY, Rouhan G, Dong SY, Liu HM, Du XY, Zhang LB, et al. A revised classification of Dryopteridaceae based on plastome phylogenomics and morphological evidence, with the description of a new genus, Pseudarachniodes. Plant Divers. 2024;47(1):34-52.
[Crossref] [Google Scholar] [PubMed]
- Choudhary CK, Fakana ST, Mengist AB. Conservation of Majang forest biosphere reserve: An opportunity through community-based ecotourism programme in Majang Zone, Gambella, South West Ethiopia. Asian J Conserv Biol. 2003;10:280-96.
- Demelash T, Kifle B. A review of coffee diseases research in Ethiopia. Int J Agric Biosci. 2015;7:65-70.
- Getaneh A, Adugna G, Alamerew S. Heterosis combining ability and heritability for resistance to coffee wilt disease in Arabica coffee. Ethiop J Agric Sci. 2001;31:19-43.
Citation: Zhang LB (2025) New Progress in the Phylogeny and Classification of the Largest Fern Family Dryopteridaceae. J Plant Pathol Microbiol. 16:762.
Copyright: © 2025 Zhang LB. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.