Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Review Article - (2025) Volume 16, Issue 6
Efficacy and safety of four-factor versus three-factor prothrombin complex concentrate for warfarin reversal in emergency haemorrhage: A systematic review and meta-analysis
Molly King and Denise E. Jackson*Received: 08-Dec-2025, Manuscript No. JBDT-25-30620; Editor assigned: 10-Dec-2025, Pre QC No. JBDT-25-30620 (PQ); Reviewed: 24-Dec-2025, QC No. JBDT-25-30620; Revised: 31-Dec-2025, Manuscript No. JBDT-25-30620 (R); Published: 07-Jan-2026, DOI: 10.4172/2155-9864.25.16.635
Keywords
Blood; Haemorrhage; Deep vein thrombosis; Direct oral anticoagulants
Description
The haemostatic balance between the physiological processes of coagulation and anticoagulation is a crucial and delicate system designed to allow clot formation when needed to prevent bleeding, and prevention of inappropriate clot formation when it is not required. For some individuals predisposed to blood clots, Deep Vein Thrombosis (DVT) and strokes or have atrial fibrillation or artificial heart valves, warfarin, a competitive antagonist of VKORCl, is used to inhibit the conversion of vitamin K 2,3-epoxide back to the active vitamin K hydroquinone, and prevents the synthesis of the vitamin K dependent coagulation factors [1]. Consequently, this inhibition of the reduction of Vitamin K 2,3-epoxide leads to the decrease of clotting ability that is seen in warfarin treated patients. Since the approval of dabigatran in 2010, Direct Oral Anticoagulants (DOACs) have begun to surpass warfarin as they do not require close blood monitoring and have fewer interactions with certain food and medications. A study looking at DOAC versus warfarin use in England between 2014 to 2019 showed DOAC prescription increasing from 9% to 74% and a decline in warfarin from 91% to 26% [2]. A similar study in the US between 2011 and 2020 showed increases of DOAC use for treatment of atrial fibrillation increase from 4.7% to 47.9% and a decline in warfarin from 52.4% to 17.7% [3]. While this decreasing trend is noted, warfarin is still the preferred anticoagulant in certain patient populations, such as those with triple-positive antiphospholipid syndrome, valvular atrial fibrillation, prosthetic cardiac valves, VTE treatment (+/- cancer) and a-fib with moderate to severe mitral stenosis or mechanical heart valves [4]. This is why, although there is an overall decrease in usage, emergency correction of warfarin-induced anticoagulation remains an important medical issue.
Current reversal strategies
Bleeding in patients on warfarin can be life threatening. The 30-day mortality of presentations to emergency with warfarin associated major bleeding is 1 in 10, with intracranial haemorrhage resulting in the largest number of associated deaths, closely followed by gastrointestinal bleeds [5]. In these situations, prompt reversal is required and are monitored by the International Normalised Ratio (INR). Though there are no official guidelines that define the ideal target INR, research indicates that most studies aim for a post-reversal INR of 1.5 and 1.3. There are a variety of methods of reversal based on severity of bleeding and include vitamin K replacement, use of Fresh Frozen Plasma (FFP) to replace coagulation factors, recombinant factor VII and the use of Prothrombin Complex Concentrates (PCCs).
In emergent haemorrhage situations, the preferred method of reversal is either FFP or PCCs. Studies comparing FFP to PCCs have shown that, in comparison to FFP, PCCs have a significantly more rapid INR correction in a significantly shorter time frame [6]. PCCs achieve reversal by replacement of coagulation factors and, additionally, are preferred over FFP due to their small volume, minimisation of viral transmission through viral inactivation and reduced risk of clinically adverse outcomes such as Transfusion Associated Circulatory Overload [7]. By bypassing their synthesis and directly supplying the vitamin K dependent coagulation factors into the blood, the blocking VKORCl by warfarin is rendered obsolete and the clotting cascade can be activated to inhibit haemorrhage.
Four-factor PCC compared to three-factor PCC
There are currently two types of PCC on the market that are utilised in the reversal of warfarin: Three-factor PCC and four-factor PCC. Prior to the FDA approval and implementation of four-factor PCC in the United States of America in 2013 and in the European and Canadian market in 1996, three-factor PCC was used off-label in America, Canada and Europe for the emergency reversal of warfarin with no specific dosing guidelines available [8], while in Australia warfarin reversal using three-factor PCC is approved.
Though both three-factor and four-factor PCC contain high concentrations of the vitamin K dependent coagulation factors II, IX, and X, four-factor PCC additionally contains VII, as well as the antithrombotic protein S and protein C [7]. With administration of three-factor PCC, it is recommended to administer FFP concurrently to see an increase in factor VII, which is not required when four-factor PCC is administered [9].
Scope of review
Current literature focuses on dosage regimes of three-factor PCC or four-factor PCC independently without comparison between each, but few have compared their efficacy and safety directly. There is a need for a systematic review and meta-analysis to assess whether there are better patient outcomes when four-factor PCC is administered over three-factor PCC, and whether there is any change in adverse outcomes. This review will aim to evaluate the differences in current literature comparing four-factor PCC and three-factor PCC in the level of successful INR reduction. Additionally, literature shows that warfarin reversal with PCCs has been associated with thromboembolic events, but it is thought that the inclusion of protein C and S in four-factor PCC acts as a protective agent against thrombosis. With this in mind, we aim to evaluate adverse thromboembolic outcomes experienced by patients and compare if there is statistical significance between three-factor PCC or four-factor PCC and thromboembolic events post treatment [10].
Rationale for meta-analysis
While current guidelines recommend four-factor PCC over three-factor PCC for the reversal of warfarin, there is a noticeable lack of research comparing the efficacy or the adverse outcomes of the two directly [11]. The variation in INR correction strategies, dosing regimens, on- and off-label use, and the selection of either three-factor PCC or four-factor PCC are all areas of debate and, coupled with inconsistencies across published studies in terms of safety and clinical efficacy between the two options, a meta-analysis of the available evidence is warranted. The Patient or Problem, Intervention, Comparison and Outcome (PICO) framework facilitated the construction of the primary research question: in patients who require emergent warfarin reversal due to haemorrhage (population), does four-factor PCC (intervention) reduce INR quicker with less adverse effects (outcome) when compared to the use of three-factor PCC (comparison)? This study aims to systematically address this research question by comparing the level of INR reversal and thrombotic adverse effects (DVT, Pulmonary Embolism (PE), Myocardial Infarction (MI), stroke, e.g.,) mortality, following infusion for emergent warfarin reversal of 3-factor PCC versus 4-factor PCC.
Materials and Methods
Study design
This systematic review was conducted following PRISMA guidelines to obtain relevant articles that directly compare three-factor PCC and four-factor PCC, and whether there are any thromboembolic events post-use [12].
Search strategy
Searches for eligible literature were conducted through the following databases: PubMed, Scopus, Google Scholar and Embase. Key search terms included “4F-PCC” AND “3F-PCC”, “4-factor prothrombin complex concentrate” AND “3-factor prothrombin complex concentrate”, “Beriplex”, “Prothrombinex”, “Vitamin K dependent concentrate”, “prothrombin complex concentrates” and “prothrombin complex concentrates comparison.” To ensure complete coverage of all terms used, both the numerical symbol and word for numbers were used when relevant. These searches were conducted without restrictions on publication date. No articles were added to the collection through manual search. Articles were retrieved from their respective database and saved into EndNote.
Eligibility criteria
From EndNote, the collection was imported into Covidence, where duplicates were removed. Articles were excluded based on title and abstract screening, then assessed for eligibility. Articles were considered eligible if they offered a direct comparison in clinical outcomes when three-factor PCC and four-factor PCC were used, the patients were being treated with warfarin, and if they provided insight into adverse thromboembolic events post-PCC use. Observational studies, including both prospective and retrospective cohort studies, were considered eligible for inclusion. Articles were excluded from the collection if they were irrelevant to the research question, systematic review/meta-analysis or review, did not offer a direct comparison between three-factor PCC and four-factor PCC, if the comparator was to FFP, or if they lacked specification of the PCC used. Additionally, studies inaccessible to the public, conference abstracts, letters, and short communications on this topic were excluded.
Participants, interventions and comparators
The “Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)” checklist was used to assess the quality of the eligible studies [13].
Outcomes
Studies that provided pre-treatment INR, post-treatment INR from both three-factor PCC and four-factor PCC arms were considered eligible for inclusion. Studies were required to outline that the patients were pre-treated with warfarin and experiencing a haemorrhage, requiring warfarin reversal. Additionally, studies were eligible for inclusion if they reported thromboembolic events post-treatment with PCCs and reported mortality events for each treatment group.
Data extraction
Data was extracted from the eligible studies and included the primary author, publication year, study design, study period, country of study, sample size, and the parameters measured in each study. The parameters that were used for this meta-analysis were the number of participants, the mean pre-treatment INR, the mean post-treatment INR, the mean change in INR and the number of adverse effects and mortality events for both the three-factor and four-factor PCC groups.
Statistical analysis
To conduct this meta-analysis, ReviewMan software was downloaded from the Cochrane Website [14]. For the INR reversal investigation, a comparison of the change in means calculated from published mean pre- and post-INR results for each article was used. For the inquiry into thromboembolic events and mortality events, a risk difference analysis was used. The data for each were presented as a forest plot, which included the overall P-value, 95% confidence intervals and heterogeneity scores. For this meta-analysis, a P-value<0.05 is considered statistically significant.
Results
Study selection
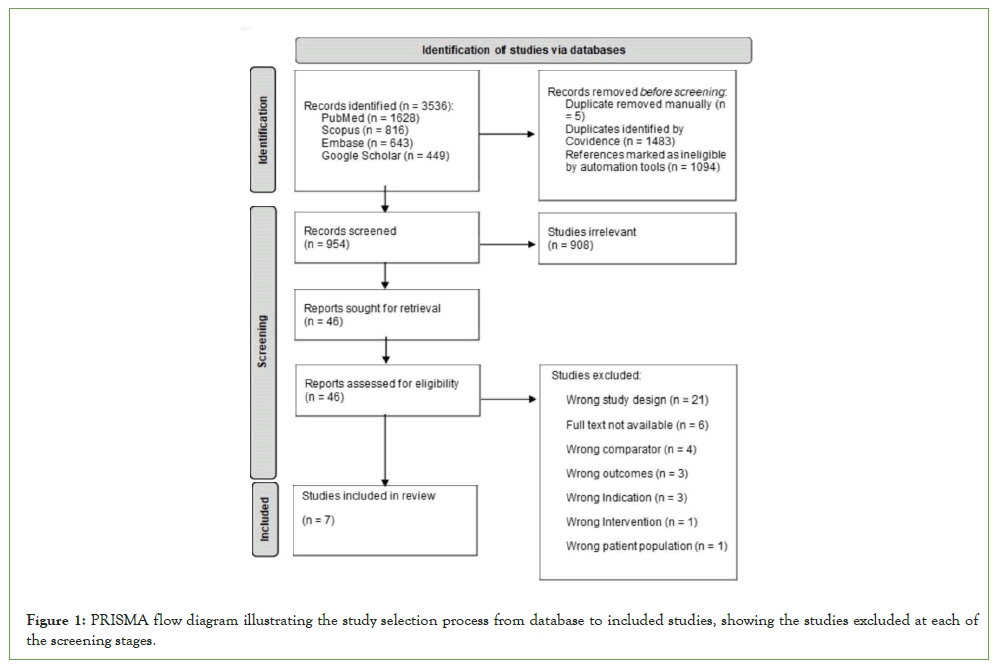
The study selection process is outlined in (Figure 1). The database search revealed 3536 potential studies (Pubmed, n=1628; Scopus, n=816; Embase, n=643; Google Scholar, n=449). Duplicates (manually and automatically) and ineligible studies identified by automation tools were removed by Covidence software, leaving 954 studies. Title and abstract screening then removed 908 studies considered irrelevant to the research question, leaving 46 for retrieval. All 46 studies were retrieved, of which 39 were removed due to incorrect study design, lack of full text, incorrect comparator group, incorrect outcomes measured, incorrect indication, incorrect intervention, or incorrect patient population. A total of 7 studies matched the eligibility criteria and were included in the meta-analysis.
Figure 1: PRISMA flow diagram illustrating the study selection process from database to included studies, showing the studies excluded at each of the screening stages.
Characteristics of included studies
The characteristics of the seven included studies are summarised in (Table 1). All seven studies in this meta-analysis are retrospective in design, and all gathered data from the United States of America. The study periods of the included studies ranged from August 2007 to August 2016. All included studies’ primary outcome was the level of INR reversal, with a goal ranging between 1.3-1.5, after treatment with either three-factor or four-factor PCC in patients who are being treated with warfarin, with six also focusing on thromboembolic effects. Sample sizes of the included studies ranged from 53 to 195 [15-17].
| Primary author | Study design | Country | Study period | Sample size total | Sample size included (excluded) | Primary outcome measured | Other outcomes measured |
|---|---|---|---|---|---|---|---|
| Al-Majzoub, et al. 2016 [9] | Single centre, retrospective cohort analysis | United States of America | August 2012 to January 2013 for 3-PCC, August 2013 to January 2014 for 4-PCC | 91 | 53 (38) | Warfarin reversal defined as INR of 1.3 or less | Change in INR from pretreatment INR, survival to hospital discharge |
| Barton, et al. 2018 [18] | Single-site, retrospective cohort study | United States of America | March 2011 through to August 2016 | 565 | 195 (370) | Efficacy at reversing INR to <1.4 upon repeat INR check after PCC admission | Complications post-reversal |
| Jones, et al. 2016 [8] | Multicentre, retrospective, propensity-matched pilot Study | United States of America | January 1 2012 through to April 15 2015 | 248 | 148 (100) | Percent of patients achieving INR equal to or less than 1.4 at initial follow up INR after PCC admission. | Thrombotic events post treatment |
| Kuroski, et al. 2017 [15] | Single centre, retrospective cohort study | United States of America | January 1st 2013 through to 31 may 2014 for 3F-PCC, June 1 2014 through to September 15 2015 for 4F-PCC | 144 | 137 (7) | Percentage of patients achieving an INR reversed to equal to or less than 1.5 within 8 hours of PCC administration | Thromboembolic events within 7 days post-PCC (incidence), all-cause mortality |
| Mangram, et al. 2016 [16] | Retrospective study | United States of America | January 2010 through to October 2014 | 64 | 64 (0) | Successful INR reversal defined as less than 1.5 post PCC | Adverse treatment effects (any thromboembolic complication). |
| Margraf, et al. 2020 [17] | Retrospective cohort study | United States of America | August 29, 2007 through to June 30, 2014 | 171 | 80 (91) | The goal INR of equal to or less than 1.5 after administration of PCC3 or PCC4. | Thromboembolic Events (TE), death during hospital stay |
| Holt, et al. 2018 [19] | Multicentre, retrospective cohort study | United States of America | May 2011 through to October 2014 | 134 | 134 (0) | Compare three-factor PCC versus 4 factor PCC in patients on warfarin | Mortality (incidence), new thromboembolism within 7 days post-pcc |
Table 1: Overview of eligible study characteristics of the seven studies included in this review.
Assessment of study quality
The seven included studies were assessed using the STROBE checklist, as shown in (Table 2). The selected STROBE criteria were mainly fulfilled. Noticeably, the majority of the studies did not explain how they arrived at their study size [8,9,15-19].
| Study | Key elements of study design early | Describes settings, locations, relevant dates, follow-up and data collection | Explain how the study size was arrived at | Report number of individuals at each stage of the study | Give reasons for non-participation at each stage | Report numbers for outcome events or summary measures | Summarise key results with reference to study objectives | Discuss limitations of the study, considering sources of potential bias or imprecision | Give the source of funding and the role of funders present in the study. |
|---|---|---|---|---|---|---|---|---|---|
| Al-Majzoub, et al. 2016 [9] | Y | Y | N | Y | Y | Y | Y | Y* | Y |
| Barton, et al. 2018 [18] | Y | Y | N | Y | Y | Y | Y | Y | Y |
| Jones, et al. 2016 [8] | Y | Y | N | Y | Y | Y | Y | Y | Y |
| Kuroski, et al. 2017 [15] | Y | Y | N | Y | Y | Y | Y | Y | N |
| Mangram, et al. 2016 [16] | Y | Y | N | Y | Y | Y | Y | Y | Y |
| Margraf, et al. 2020 [17] | Y | Y | N | Y | Y | Y | Y | Y | Y |
| Holt, et al. 2018 [19] | Y | Y | Y | N | N | Y | Y | Y | Y |
Table 2: Evaluation of eligible studies as per the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist.
Meta-analysis
Data extracted from the eligible studies are included in (Table 3). For studies that presented data as median and IQR, estimates of the mean and standard deviation were calculated and reported in (Table 3) [13]. This data was used to perform the meta-analysis, resulting in forest plots for the mean change in INR pre- and post-treatment with either three-factor or four-factor PCC, and for thromboembolic adverse effects experienced post-warfarin reversal with either three-factor or four-factor PCC. Mortality events were presented as the number of mortality events over the total of participants in each group. These forest plots, along with the risk of bias, are shown in (Figure 2).
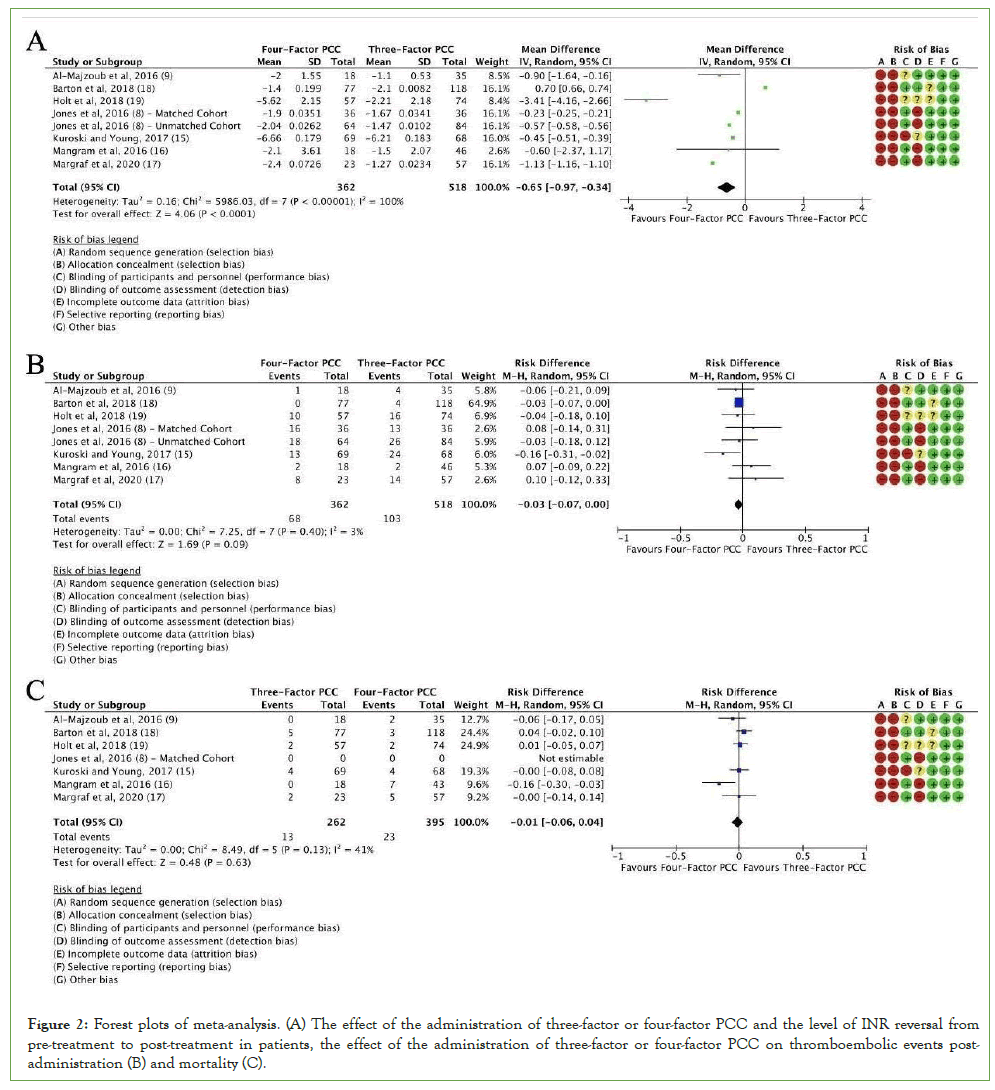
Figure 2: Forest plots of meta-analysis. (A) The effect of the administration of three-factor or four-factor PCC and the level of INR reversal from pre-treatment to post-treatment in patients, the effect of the administration of three-factor or four-factor PCC on thromboembolic events post-administration (B) and mortality (C).
INR reversal
In this meta-analysis, 515 patients were treated with three-factor PCC, and 362 patients received four-factor PCC. The overall mean difference in INR was -0.65 with a 95% CI [-0.97, -0.34] with a P=0.0001 (Figure 2A). This demonstrates a statistically significant reduction in INR, favouring four-factor PCC. These findings suggest that the use of four-factor PCC for emergent warfarin reversal provides a more rapid and effective reversal when compared with three-factor PCC. However, the heterogeneity of the analysis is exceptionally high, with an I2=100%.
Incidence of thromboembolic events
The assessment of thromboembolic outcomes revealed that 23 of the 396 patients in the three-factor PCC group experienced a post-treatment thromboembolic event, compared with 13 of 262 patients in the four-factor PCC cohort (Figure 2B). The calculated Risk Difference (RD) was -0.1, indicating a slight reduction in risk with the use of the four-factor PCC. The 95% CI is [-0.06, 0.04], which encompasses the null value; therefore, this difference is deemed not statistically significant. This finding suggests that the use of four-factor PCC does not significantly increase or decrease the risk of post-treatment thromboembolic events relative to three-factor PCC, or vice versa, as the difference in risk is only 1%. The heterogeneity score, I2, is 0.41, indicating that 41% of the variation in the results is due to differences across studies.
Incidence of mortality outcomes
The assessment of mortality outcomes revealed that 103 deaths occurred in the three-factor PCC and 68 deaths occurred in the four-factor PCC group (Figure 2C). The calculated Risk Difference (RD) for comparing mortality between the four-factor PCC and three-factor PCC groups was -0.03, indicating a slight reduction in mortality with the use of four-factor PCC. The 95% CI is [-0.07, 0.00], which does not encompass the null value; therefore, the difference is not deemed statistically significant. Reinforcing this, the diamond touches the line of no effect, further indicating that the result is not statistically significant. The heterogeneity of these results, I2, was very low at 3%, indicating that the results across studies were highly similar.
Discussion
This meta-analysis evaluated whether four-factor PCC is clinically superior to three-factor PCC for warfarin reversal in emergency haemorrhage situations. There was a statistically significant difference in INR reversal, favouring the use of four-factor PCC. The difference in thromboembolic events or mortality between the two groups were not statistically significant and showed no superiority to one group.
INR reversal
The INR reversal suggests substantial variation in the effect sizes in this study. Contributing factors likely include differences in patient populations across the included studies, variation in baseline INR, natural variation in patients, and discrepancies in dosing strategies between three-factor and four-factor PCC and between hospitals and studies. Despite this variability, the consistent direction of effect across studies indicates that four-factor PCC is more effective for achieving greater INR reversal in emergent warfarin reversal situations than three-factor PCC.
Incidence of thromboembolic events
The study by Mangram et al had the highest number of adverse effects among the included studies for the three-factor PCC, while also reporting the fewest for the four-factor PCC [16]. Barton et al is the only study to report more adverse effects in the four-factor PCC group than in the three-factor PCC group, whereas all other studies showed a slight decrease. Overall, the majority of the included studies show a similar incidence of thromboembolic in each of the three-factor PCC and four-factor PCC groups. Previous literature had hypothesised that the four-factor PCC may have protective effects against adverse thromboembolic events due to its inclusion of factor VII and the antithrombotic proteins S and C. This hypothesis was not seen in this current study.
Incidence of mortality outcomes
Overall, the meta-analysis result suggest that there is no statistically significant difference in mortality events between the four-factor PCC and three-factor PCC groups. It is worth noting that detailed reporting on mortality was limited across the studies, with only Margraf et al providing details into the deaths, attributing all reported deaths to bleeding, likely due to inadequate warfarin reversal while haemorrhaging.
Comparison with prior meta-analyses
Through database searching, two previous meta-analyses comparing three-factor PCC and four-factor PCC in warfarin reversal were identified. The analysis by Voils and Baird published in 2014, which I believe to be the first published meta-analysis on this topic, evaluated the proportion of patients that achieved an INR ≤ 1.5 within one hour of PCC administration [20]. However, it did not assess or address mortality or thromboembolic events. This meta-analysis concluded that four-factor PCC was more effective at achieving a rapid INR correction when compared with three-factor PCC. The analysis by Margraf et al published in 2024 examined the effectiveness of their included studies in achieving the study-defined INR goal following the administration of either three-factor or four-factor PCC [21]. This study also included secondary outcomes examining thromboembolic events and survival during the hospital stay. The primary and secondary outcomes of this analysis were similar to those discussed in this study. However, this study assessed mean INR reversal rather than whether INR reversal to their predetermined INR level was achieved, and it focused on patient survival, whereas this study focuses on mortality. Additionally, this meta-analysis is similar to the reporting in the current study, which also used forest plots with heterogeneity measures. The analysis by Margraf et al concludes that the use of four-factor PCC for warfarin reversal increased patients' odds of achieving the target INR by three times compared with those who received three-factor PCC. The secondary outcomes align with the current study as there was no statistically significant difference in thromboembolic events or hospital survival rates [21].
Limitations
This study was not without limitations. One major limitation of this meta-analysis is the inclusion of all retrospective studies, which are inherently prone to selection bias, as the allocation of treatment groups and patient inclusion in the study are determined by clinical judgement rather than random allocation. Additionally, these studies rely on existing medical records, of which may be incomplete, inconsistent or contain missing data. Furthermore, retrospective studies tend to have different study designs, patient populations and outcome/endpoint definitions. These differences likely contributed to the heterogeneity score in this meta-analysis. The absence of prospective trials increases the risk of publication bias, as studies with inconclusive or negative results may not have been reported. Additionally, unequal sample sizes and mortality outcomes that were not characterised in detail were present. For instance, Holt, et al. calculated that a total of 266 patients (133 per arm of the study) would be required to achieve statistical significance; however, the study ultimately only included 124 patients (77 and 57 per group).
Conclusion
This systematic review reveals statistically significant evidence for the use of four-factor PCC for warfarin reversal, as measured by INR correction, in emergency haemorrhage situations, compared with three-factor PCC. Four-factor PCC showed an increased level of INR reduction for warfarin reversal. The data has also revealed that there is no statistically significant difference in the level of thromboembolic events post-treatment or mortality outcomes between the use of three- factor and four-factor PCC. However, with the included studies’ smaller patient populations, there is scope for further research with larger cohorts to investigate this further.
Funding Statement
The authors received no financial support for the research.
Conflicts of Interest
No conflicts of interest have been declared.
References
- World Health Organization. The global burden of cardiovascular diseases and risk factors 1990-2021. Lancet. 2023;402(10418):1285-1398.
- Ojha N, Dhamoon AS. Myocardial infarction. InStatPearls [Internet]. 2023. StatPearls Publishing.
- Salisbury AC, Alexander KP, Reid KJ, Masoudi FA, Rathore SS, Wang TY, et al. Incidence, correlates, and outcomes of acute, hospital-acquired anemia in patients with acute myocardial infarction. Circ Cardiovasc Qual Outcomes. 2010;3(4):337-346.
- Thygesen K, Alpert JS, White HD, Jaffe AS, Apple FS, Galvani M, et al. Universal definition of myocardial infarction: Kristian Thygesen, Joseph S. Alpert and Harvey D. White on behalf of the joint ESC/ACCF/AHA/WHF task force for the redefinition of myocardial infarction. J Am Coll Cardiol. 2007;28(20):2525-2538.
- Marik PE, Corwin HL. Efficacy of red blood cell transfusion in the critically ill: A systematic review of the literature. Crit Care Med. 2008;36(9):2667-2674.
- Hébert PC, Wells G, Blajchman MA, Marshall J, Martin C, Pagliarello G, et al, Transfusion Requirements in Critical Care Investigators for the Canadian Critical Care Trials Group. A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. N Engl J Med. 1999;340(6):409-417.
- Colombo MG, Kirchberger I, Amann U, Heier M, Thilo C, Kuch B, et al. Association between admission anemia and long-term mortality in patients with acute myocardial infarction: Results from the MONICA/KORA myocardial infarction registry. BMC Cardiovasc Disord. 2018;18(1):50.
- Villanueva C, Colomo A, Bosch A, Concepción M, Hernandez-Gea V, Aracil C, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368(1):11-21.
- Carson JL, Stanworth SJ, Guyatt G, Valentine S, Dennis J, Bakhtary S, et al. Red blood cell transfusion: 2023 AABB international guidelines. JAMA. 2023;330(19):1892-1902.
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, et al. 2023 focused update of the 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2023;44(37):3627-3639.
- Corwin HL, Gettinger A, Pearl RG, Fink MP, Levy MM, Abraham E, et al. The CRIT Study: Anemia and blood transfusion in the critically ill-current clinical practice in the United States. Crit Care Med. 2004;32(1):39-52.
- Carson JL, Stanworth SJ, Roubinian N, Fergusson DA, Triulzi D, Doree C, et al. Transfusion thresholds for guiding red blood cell transfusion. Cochrane Database Syst Rev. 2021;12(12):CD002042.
- DeFilippis AP, Abbott JD, Herbert BM, Bertolet MH, Chaitman BR, White HD, et al. Restrictive versusliberal transfusion in patients with type 1 or type 2 myocardial infarction: A prespecified analysis of the MINT trial. Circulation. 2024;150(23):1826-1836.
- Cooper HA, Rao SV, Greenberg MD, Rumsey MP, McKenzie M, Alcorn KW, et al. Conservative versus liberal red cell transfusion in acute myocardial infarction (the CRIT Randomized Pilot Study). Am J Cardiol. 2011;108(8):1108-1111.
- Ducrocq G, Gonzalez-Juanatey JR, Puymirat E, Lemesle G, Cachanado M, Durand-Zaleski I, et al. Effect of a restrictive vs liberal blood transfusion strategy on major cardiovascular events among patients with acute myocardial infarction and anemia: The REALITY randomized clinical trial. JAMA. 2021;325(6):552-560.
- Gonzalez-Juanatey JR, Lemesle G, Puymirat E, Ducrocq G, Cachanado M, Arnaiz JA, et al. One-year major cardiovascular events after restrictive versus liberal blood transfusion strategy in patients with acute myocardial infarction and anemia: The REALITY randomized trial. Circulation. 2022;145(6):486-488.
- Simon T, Herbert BM, Brooks MM, Goodman SG, Alexander JH, Steg PG, et al. Restrictive or liberal transfusion strategy in patients with acute myocardial infarction and anemia: 6-month mortality in the MINT trial. Circulation. 2024;150(13):1064-1066.
- Rao SV, Jollis JG, Harrington RA, Granger CB, Newby LK, Armstrong PW, et al. Relationship of blood transfusion and clinical outcomes in patients with acute coronary syndromes. JAMA. 2004;292(13):1555-1562.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ. 2021;372.
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions version 6.3. 2022.
- Review Manager (RevMan). Version 5.4. Copenhagen: Cochrane Collaboration; 2020.
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177-188.
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560.
- Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629-634.
- Carson JL, Brooks MM, Hébert PC, Goodman SG, Bertolet M, Glynn SA, et al. Restrictive or liberal transfusion strategy in myocardial infarction and anemia. N Engl J Med. 2023;389(26):2446-2456.
- Donadee C, Raat NJ, Kanias T, Tejero J, Lee JS, Kelley EE, et al. Nitric oxide scavenging by red blood cell microparticles and cell-free hemoglobin as a mechanism for the red cell storage lesion. Circulation. 2011;124(4):465-476.
- Piagnerelli M, Boudjeltia KZ, Vanhaeverbeek M, Vincent JL. Red blood cell rheology in sepsis. Intensive Care Med. 2009:273-282.
- Hod EA, Zhang N, Sokol SA, Wojczyk BS, Francis RO, Ansaldi D, et al. Transfusion of red blood cells after prolonged storage produces harmful effects that are mediated by iron and inflammation. Blood. 2010;115(21):4284-4292.
- Weinberg JA, Patel RP. Red blood cell transfusion in the ICU: Mechanisms and storage lesions. Curr Opin Anaesthesiol. 2016;29(2):198-204.
- Tsai AG, Johnson PC, Intaglietta M. Oxygen supply through microvascular networks. Transfusion. 2010;50(5):1040-1049.
- Chien S, Dormandy J, Ernst E, Matrai A. Summary, Conclusions and Perspectives. InClinical Hemorheology: Applications in Cardiovascular and Hematological Disease, Diabetes, Surgery and Gynecology. 1987;375-381. Dordrecht: Springer Netherlands.
- Tinmouth A, Fergusson D, Yee IC, Hébert PC, ABLE Investigators and the Canadian Critical Care Trials Group. Clinical consequences of red cell storage in the critically ill. Transfusion. 2006;46(11):2014-2027.
- Vallet B, Robin E, Lebuffe G. Venous oxygen saturation as a physiologic transfusion trigger. Crit Care. 2010;14(2):213.
- Mazin Ali. Image captured using Bio-Rad ChemiDoc XRS+ and Image Lab software. Bio-Rad Laboratories. 2025.
- Shander A, Javidroozi M. Anemia and red blood cell transfusion in the adult cardiac surgery patient: A review of the literature. Cureus. 2021;13(8):e17096.
- Arynov A, Kaidarova D, Kabon B. Alternative blood transfusion triggers: A narrative review. BMC Anesthesiol. 2024;24(1):71.
- DeFilippis AP, Blaha MJ. Defining myocardial infarction: Grades of severity or different aetiology. Eur Heart J Acute Cardiovasc Care. 2025;14(3):142-144.
- Murphy EL, Kwaan N, Looney MR, Gajic O, Hubmayr RD, Gropper MA, et al. Risk factors and outcomes in transfusion-associated circulatory overload. Am J Med. 2013;126(4):357-e29.
- Carson JL, Guyatt G, Heddle NM, Grossman BJ, Cohn CS, Fung MK, et al. Clinical practice guidelines from the AABB: Red blood cell transfusion thresholds and storage. JAMA. 2016;316(19):2025-2035.
- Xu C, Li X, Wang L, Zhang Y, Liu J. Red blood cell transfusion strategy and clinical outcomes in myocardial infarction: A systematic review and meta-analysis. iScience. 2023;26(8):107429.
Citation: King M, Jackson DE. (2025). Efficacy and Safety of Four-Factor versus Three-Factor Prothrombin Complex Concentrate for Warfarin Reversal in Emergency Haemorrhage: A Systematic Review and Meta-Analysis. 16:635.
Copyright: © 2025 King M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.