Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Proquest Summons
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Review Article - (2025) Volume 16, Issue 6
Effectiveness of NIPT on RhD-negative pregnant women on RhIg-induced isoimmunisation and associated healthcare costs: systematic review and meta-analysis
Pooja C Peiris1, Cody Smith1, Habtom Makel1 and Denise E. Jackson1,2*2Department of Thrombosis and Vascular Diseases Laboratory, School of Health and Biomedical Sciences, RMIT University, Victoria, Australia
Received: 24-Nov-2025, Manuscript No. JBDT-25-30368; Editor assigned: 26-Nov-2025, Pre QC No. JBDT-25-30368 (PQ); Reviewed: 10-Dec-2025, QC No. JBDT-25-30368; Revised: 17-Dec-2025, Manuscript No. JBDT-25-30368 (R); Published: 24-Dec-2025, DOI: 10.4172/2155-9864.25.16.634
Keywords
NIPT; Alloimmunisation; RhIg prophylaxis; Cost; RhD-negative pregnant women.
Introduction
Background
Over centuries, Rhesus D antigen (RhD) alloimmunisation has been a common concern in obstetric studies for pregnant women in the quest to eliminate the risk of Haemolytic Disease of the Foetus and Newborn (HDFN) [1]. In routine care, antibody screening, invasive diagnosis of the foetus, and administration of anti-D prophylaxis are carried out with close monitoring of the mother and foetus to minimise HDFN in RhD- negative women. Yet, invasive procedures, including Chorionic Villus Sampling (CVS) and amniocentesis, possess a risk of provoking spontaneous miscarriage with a 0.5-1% degree of risk [2,3]. Antibody screening functions as an indirect method for assessing foetal status. However, their sensitivity is lower compared to invasive techniques, highlighting the need for non-invasive diagnostic methods [4]. Thus, in recent years, there has been a shift towards foetal RhD genotyping using Cell-Free DNA (cfDNA) to guide Rh Immunoglobulin (RhIg) usage in pregnant women and to enhance foetal survival. Conversely, the cost and complexity of these tests have impeded the widespread adoption of routine Non-Invasive Prenatal Testing (NIPT) for foetal RhD prediction and the determination of RhIg eligibility [5].
Use of RhIg and associated risk in pregnancy
The development and introduction of postnatal RhIg treatment in the 1960s led to a significant reduction in the prevalence of HDFN due to Rh incompatibility, from 16% to 2% [5,6]. These figures were further reduced to 0.1%-0.3% with prenatal prophylaxis at a gestational age of 28-34 weeks in the 1970s [6]. Anti-D immunoglobulin is a specific biological product obtained primarily through plasma apheresis from volunteers with strong anti-D antibody titres [6]. This influences the availability of RhIg doses, which are typically limited [6,7]. Many countries rely on importing RhIg due to its non-local production, unavailability of raw materials, or donor shortages [6-8]. Consequently, guidelines for the use of anti-D prophylaxis have been established and vary between countries, particularly regarding the frequency and dosage of both prenatal and postnatal administration [2]. Additionally, application of RhIg increases the chance of disease transmission. Although blood products are screened to minimise the risk, subsequent studies have revealed the existence of tainted RhIg batches containing viruses or prions, such as the Hepatitis C virus [6-8]. In the process of RhIg production, Volunteers being immunised against the RhD antigen also subject themselves to a greater risk in future transfusions. This raises ethical concerns about RhIg use, prompting interest in targeted RhIg prophylaxis [5,7,9]. RhIg is relatively expensive due to limited resources and requires optimised management to conserve supply and minimise excessive utilisation. In RhD-negative pregnancies, about 40% of women are at no risk of alloimmunisation [5,10]. Hence, predetermination of foetal RhD status in advance could avoid treatment involving an RhD- negative foetus, including intensive care or RhIg administration [9].
NIPT for foetal RhD detection in prenatal care
Prenatal cfDNA or NIPT is used to efficiently predict foetal RhD status and target RhIg administration in all RhD-negative, non- alloimmunised pregnant women, enabling accurate detection of RhD- positive foetuses and reducing unnecessary RhIg use [6,8,11]. As a result, this approach has been adopted into routine clinical practice in many European countries, enhancing the management of RhD- negative pregnant women [4,11]. Cell-Free DNA (cfDNA) is present at approximately 10% of the total maternal plasma concentration from 10 to 12 weeks of gestation, increases with gestational age, and rapidly disappears from the maternal circulation postpartum [4]. To accurately predict foetal DNA status using cfDNA, it is essential to consider genetic variants, such as point mutations that cause weak D types, which may hinder estimation of the foetal RhD status [10,12]. In Caucasians, the RhD-negative phenotype is often caused by RhD gene deletion, whereas in individuals of African origin, it is typically caused by the RHD Pseudogene (RhDΨ) or the RHD-CE-DS (dCeS) haplotype [5,13,14]. Therefore, the frequencies of RHD variants in different populations should be considered when selecting gene targets. Consequently, multiple combinations including RhD exons 5, 7, and 10 are commonly used as the choice of exon targets [10,12]. By targeting multiple exons in a single assay, high accuracy can be achieved [12]. It is crucial to acquire a high sensitivity during RHD screening assays, as a false negative result may result in omitting anti-D administration, leaving the pregnant woman at risk of sensitisation against anti-D [10].
Scope of the review
In standard care, routine antenatal RhD screening is utilised to identify maternal pregnancies at risk of HDFN, followed by prenatal and postnatal RhIg therapy based on the foetal RhD status in RhD-negative pregnant women [1]. Despite precautions with anti-D prophylaxis administration, residual cases of RhD alloimmunisation still occur in RhD-negative pregnant women [13]. Therefore, using NIPT for foetal RhD identification from maternal plasma enables the withholding of anti-D prophylaxis from RhD-negative pregnant women carrying RhD- negative foetuses, preventing unnecessary administration of RhIg and reducing the risk of alloimmunisation [2,13].
Most investigations in RhD genotyping have employed labour- intensive and extravagant approaches, which are incompatible with large-scale screening. The advent of robotic technologies for isolating foetal DNA and their adaptation to prenatal testing permitted high- throughput analysis, delivering an efficient and cost-effective approach to avoiding the regular administration of RhIg to all RhD-negative pregnant mothers [3].
Consequently, a systematic review and meta-analysis of existing data would be valuable in assessing the impact of NIPT on RhIg-induced isoimmunisation and related healthcare expenses.
The study aims to utilise the PICO framework to compare current data and address the following research question: Does NIPT-guided anti-D prophylaxis (intervention) reduce maternal sensitisation episodes (outcome) in RhD-negative pregnant women (population), relative to universal anti-D prophylaxis (comparison)? A secondary question aims to address: Does employing non-invasive RhD typing (intervention) for RhIg administration mitigate healthcare costs (outcome) when compared to routine RhIg (comparator) in RhD-negative pregnant women (population)?
Methods
Study design
The Preferred Reporting Items for Systematic Reviews and Meta- Analyses (PRISMA) guidelines were followed in conducting the systematic review to obtain specific studies that investigate the NIPT efficacy in minimising the incidence of Rh-D sensitisation with RhIg administration and associated costs [15].
Search strategy
Database searches were conducted through PubMed, Scopus, Embase, and Google Scholar, with no restrictions on publication dates, to identify relevant literature. "NIPT," "non-invasive prenatal testing," "cell-free DNA," "cfDNA," "Rh-D genotyping," "RhIg prophylaxis," "anti-D prophylaxis," and "Rh-D negative pregnant women" were key search terms included. To ensure complete coverage, both British and American spellings were incorporated where appropriate. No articles were manually added through searches. EndNote was used to save the articles that were found through database searches.
Eligibility criteria
Articles were arranged for eligibility assessment using EndNote [16]. Using Covidence, duplicates were removed, and the process of including and excluding articles was initiated [17]. Before being assessed for eligibility, articles were first excluded based on their abstract and title. Those that examined the risk or incidence of RhIg-induced alloimmunisation, and healthcare costs associated were considered eligible. Case studies, systematic reviews, meta-analyses, abstracts only, and articles that were not relevant to the research issue or that were not written in English were all excluded.
Methodology quality assessment
Using the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) criteria, the methodological quality of the eligible studies was assessed [18].
Data management and extraction
Primary author, research design, country of publication, study period, sample size, and parameters assessed for analysis were among the data collected from eligible studies. The selected characteristics comprised sensitisation events and the costs related to target and routine anti-D prophylaxis.
Statistical analysis
RevMan (Review Manager) software (Version 5.4.1, The Cochrane Collaboration, and UK) was used to perform the meta-analysis. The frequency of sensitisation events along with costs associated with the targeted anti-D versus routine anti-D prophylactic groups were compared using a two-way proportional analysis.
When Standard Deviations (SDs) for cost data were not reported, we estimated them using an assumed Coefficient of Variation (CV). Following methods commonly employed in health economic evaluations (e.g. Weir, et al. 2018), we adopted a base CV of 0.5. As the costs were presented in different currencies, all values were converted to Australian Dollars (AUD) for analysis (AUD 1$=0.56 euro/ 0.91 CAD/0.66 USD).
Sensitisation events were analysed using the Mantel-Haenszel method, and a random-effects model was employed to compute the Odds Ratio (OR). A random-effects model was used to analyse costs and determine the standardised mean difference using the Inverse Variance approach. Forest plots were used to illustrate the results for each parameter. In addition to the 95% CI and I2 statistic to examine research heterogeneity, RevMan software computed the total P-value to determine statistical significance. Further, associated heterogeneity was taken into account. Statistical significance is defined as a P-value of less than 0.05.
Using the Risk Of Bias In Non-randomised Studies of Interventions (ROBINS-I) tool, the risk of bias for the studies was evaluated [19]. Each study was independently reviewed and assigned a risk level (low, unclear, high) across these domains [20-23].
Results
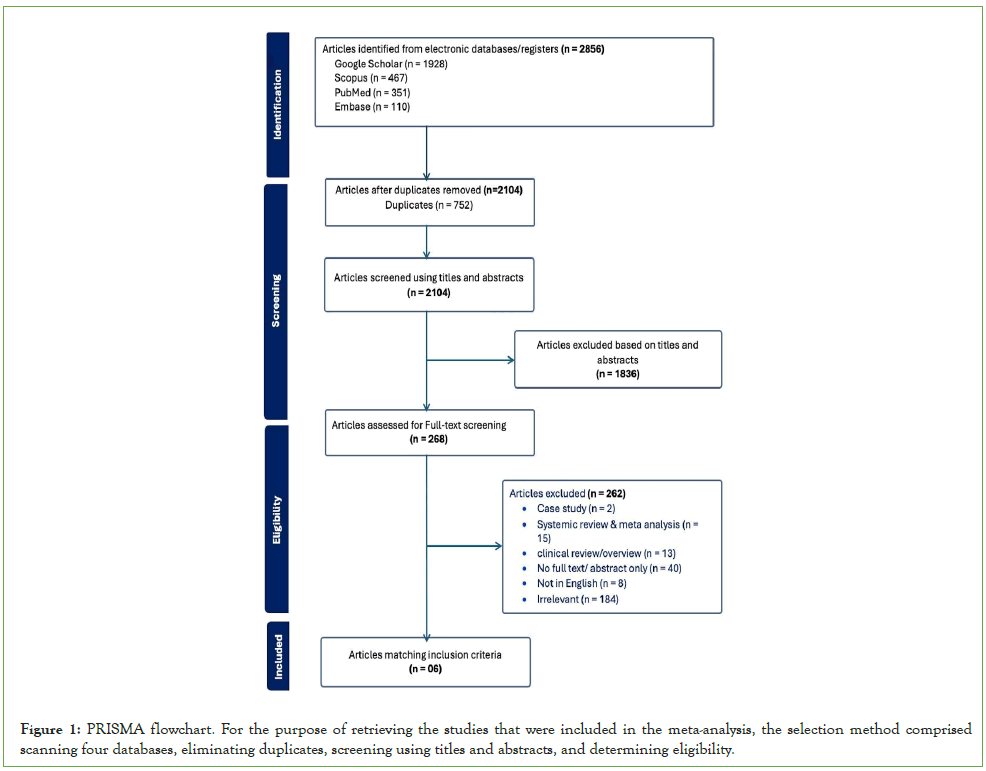
The inclusion and exclusion criteria displayed served as the basis for the study selection (Figure 1). A total of 2856 possibly pertinent studies were found in the database search [24,25].After identifying and eliminating duplicates, 2104 studies remained. 1,836 of these studies were excluded after initial filtering based on their titles and abstracts [26-28].The eligibility of the remaining 268 studies was then evaluated. The primary exclusion criterion, which led to the elimination of 184 papers, was insignificant to the investigated question. Similarly exempt were case studies, abstract-only papers, review articles, and publications not in English. The meta-analysis comprised six papers in total that fulfilled the qualifying requirements [29,30].
Figure 1: PRISMA flowchart. For the purpose of retrieving the studies that were included in the meta-analysis, the selection method comprised scanning four databases, eliminating duplicates, screening using titles and abstracts, and determining eligibility.
Study characteristics
The characteristics of the six studies that assessed the effects of NIPT on reducing RhIg-associated sensitisation events, and healthcare costs are shown in (Table 1). Among the studies incorporated, four emphasised the incidence of isoimmunisation, and four focused on the healthcare costs involved per pregnancy following RhIg administration. The studies conducted spanned mostly over European countries, which were either retrospective, prospective, or model-based analyses. The total number of pregnant women who are Rh-D-negative comprises the sample size.
| Study | Study design | Country | Study period | Sample size | Parameters measured |
|---|---|---|---|---|---|
| Darlington, et al. 2018 [28] | Prospective | France | 2009-2013 | 850 | Foetal RhD status, RhIg administration, direct health costs per pregnancy |
| Neovius, et al. 2015 [29] | Model based analysis | Sweden | 2008-2011 | 13,822 | Cost per pregnancy, incidence of RhD immunisation |
| Teitelbaum, et al. 2015 [31] | Model based analysis | Canada | 2010 | 69,286 | Cost per pregnancy, RhIg doses administered, incidence of RhD immunisations |
| Tiblad, et al. 2013 [32] | Prospective | Sweden | 2009-2011 | 9,380 | Foetal RhD status, incidence of RhD immunisation |
| Demirel, et al. 2018 [27] | Retrospective | Turkey | 2011-2015 | 1135 | Cost per pregnancy |
| Thorup, et al. 2025 [20] | Retrospective | Denmark | 2004-2020 | 506 | Incidence of RhD immunisation |
Table 1: Eligible study characteristics for the NIPT effectiveness in reducing RhIg-associated sensitisation events and healthcare costs.
The study data entailed in the meta-analysis (Table 2). The proportion of total sensitisation events that occurred and the healthcare costs associated were calculated per pregnancy. When measuring the sensitisation events that occurred, the data were used over the total number of RhD-negative mothers given routine or targeted RhIg prophylaxis to determine if it impacted the incidence of alloimmunisation. The costs were measured as the average number per pregnancy, giving routine or targeted anti-D to assess cost-effectiveness.
| Study | n (total population) | RhIg dose | Week of administration | Sensitisation Routine | Sensitisation Targeted | Cost per pregnancy (AUD$) Routine | Cost per pregnancy (AUD$) Targeted |
|---|---|---|---|---|---|---|---|
| Darlington, et al. 2018 [28] | 850 | 300 μg (1500 IU) | 28 ± 1 | - | - | 804.1 | 804.1 |
| Neovius, et al. 2016 [29] | 13,882 | 250-300 μg (1250-1500 IU) | 29 | 33/7099 | 11/6723 | 2060.39 | 2060.39 |
| Teitelbaum, et al. 2015 [31] | 69,286 | 300 μg (1500 IU) | 28 | 13/10,393 | 13/10,393 | 78.51 | 78.51 |
| Tiblad, et al. 2013 [32] | 9,380 | 250-300 μg (1250-1500 IU) | 28-30 | 86/18546 | 24/9380 | - | - |
| Demirel, et al. 2018 [27] | 1,135 | - | - | - | - | 393.13 | 393.13 |
| Thorup, et al. 2025 [20] | 75,156 | 250-300 μg (1250-1500 IU) | 29 | 282/60,952 | 167/75,156 | - | - |
Table 2: Eligible study data reporting sensitisation events and costs associated with routine RhIg and targeted RhIg prophylaxis.
Assessment of methodological quality
The STROBE checklist was used to assess the methodological quality of the studies included in the systematic review (Table 3). All studies received favourable scores for providing adequate detail in the title/ abstract, introduction, findings, and discussion sections. In the methodology sections, the majority also demonstrated comparatively high quality. However, four studies merely mentioned the inclusion criteria, and only two studies described the steps used to fulfil the eligibility requirements during participant selection. The majority of the studies were of excellent methodological quality overall, as displayed which mitigates the possibility of obtaining insufficient studies for the analysis.
| Title and abstract |
Introduction | Methods | Results | Discussion | |||||
|---|---|---|---|---|---|---|---|---|---|
| Clear title and abstract with study design indicated | Explains scientific background, rationale and objectives for study | Detailed study methods given | Eligibility criteria for participant selection shown | Give sources of data and details of methods of assessment (measurement) | Describes statistical methods | Give characteristic of study participants | Report number of outcome events or summary measures over time | Summarise key results and discusses limitations | |
| Darlington, et al. 2018 [28] | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Neovius, et al. 2016 [29] | Y | Y | Y | Pa | Y | Y | Y | Y | Y |
| Teitelbaum, et al. 2015 [31] | Y | Y | Y | Pa | Y | Pb | Y | Y | Y |
| Tiblad, et al. 2013 [32] | Y | Y | Y | Pa | Y | Y | Y | Y | Y |
| Demirel, et al. 2018 [27] | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| Thorup, et al. 2025 [20] | Y | Y | Y | Y | Y | Y | Y | Y | Y |
Note: Y: Criteria fulfilled; P: Criteria partially fulfilled; a: Study exclusion criteria not described; b: Incorporated statistical methods were not explicitly mentioned.
Table 3: Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist-based methodological quality evaluation of included studies.
Meta-analysis on the proportion of RhIg-induced isoimmunisation and cost associated
The construction of a meta-analysis and a forest plot for overall sensitisation events associated with RhIg administration in RhD- negative pregnant women (Figure 2). With an overall P-value of <0.001, the two-arm proportional analysis of RhIg-induced alloimmunisation was statistically significant, as shown in part A. With an I2 value of 33%, the findings of the four included studies demonstrated heterogeneity. The I2 value shows substantial heterogeneity. As a result, the data exhibits moderate fluctuation. The findings suggest that, in comparison to routine RhIg prophylaxis, NIPT reduces RhIg-induced alloimmunisation in maternal care (95% CI, 0.39, 0.68).
Figure 2: Meta-analysis of forest plots on the effectiveness of NIPT on maternal care management in Rh-D negative pregnant women. Note: (A) The incidence of RhIg-associated alloimmunisation as a portion of the total number of pregnancies was determined using two-armed proportion analysis data; (B) Standard mean analysis was used to express the RhIg-associated cost in maternal care. Applying the random-effects model, the 95% CI was used to determine each parameter's maximum probability. The I value and its corresponding P-value were used to assess heterogeneity, while the total P-value was used to estimate statistical significance.
A meta-analysis and forest plot were conducted to determine the overall cost associated with targeted and routine maternal care in RhD- negative pregnant women, and are displayed in part B.
With an overall P-value of 0.01, the two-arm standard mean analysis on total cost was shown to be statistically significant. Based upon the findings of the four included studies, the I2 was 100% and the heterogeneity P-value was less than 0.001. This suggests high heterogeneity, which implies significant data variability. The results suggest that the mean cost associated with targeted anti-D prophylaxis is higher than that of routine RhIg administration (95% CI: 0.11, 0.80).
Discussion
Effective management of RhIg-induced alloimmunisation with NIPT
Following this meta-analysis and systematic review, the incidence of Rh alloimmunisation in pregnant women has decreased due to the integration of NIPT into patient care for RhIg administration. This is supported by statistically significant results showing that a total of 215 (0.2%) events of alloimmunisation occurred with targeted anti-D administration compared to routine, where a total of 414 (0.4%) events occurred (95% CI, 0.39, 0.68). A heterogeneity of I2=33% was observed, indicating variability in the data across studies rather than chance. These results on the incidence of RhIg alloimmunisation represent an enhancement in clinical outcomes and support the incorporation of NIPT into routine antenatal care protocols for RhD-negative mothers.
The Rh incompatibility in RhD-negative mothers carrying an Rh-positive foetus causes sensitisation during the antenatal or postnatal stage. The use of anti-D prophylaxis in clinical practice helps reduce the incidence of HDFN by preventing alloimmunisation. The use of targeted prenatal anti-D prophylaxis is becoming increasingly widespread as the foetal RhD status can be reliably and sensitively determined from maternal blood. This development has resulted in incorporating NIPT RhD genotyping into clinical setting for the targeted prevention and management of RhD-sensitised women [8,20]. A previous study by Manzanares, et al. demonstrated a 98.65% sensitivity in foetal genotyping at a median gestational age of 12 weeks, suggesting that NIPT can be used for early diagnosis and to prevent unnecessary RhIg administration [21].
In our study, the effect of targeted RhIg was clearly demonstrated by the decline in new RhD immunisation cases in the first pregnancy when comparing the two cohorts. However, prolonged transplacental haemorrhage, a higher maternal Body Mass Index (BMI), or a failure to deliver RhD immunoglobulin following potentially sensitising events or at an adequate dose might still result in anti-D sensitisation despite these immunoprophylaxis practices [22,23]. According to our study, the recommended dosage of anti-D prophylaxis varies by country, typically 250-300 μg (1250-1500 IU), and this may not always provide adequate protection, leaving the risk of alloimmunisation despite preventive measures. A study by Soothill, et al. reported that a proportion of RhD-negative women declined RhIg administration [24]. This decision could potentially result in RhD alloimmunisation if the foetus is RhD-positive. As a result, implementing NIPT to determine foetal RhD status early could raise maternal awareness and increase uptake of RhIg prophylaxis among RhD-negative women carrying an RhD-positive foetus, thereby reducing the possibility of alloimmunisation. Moreover, using the NIPT guide management not only enhances prophylaxis administration but also builds trust and encourages engagement between patients and healthcare providers. By providing a clear, personalised understanding of the foetal RhD status, pregnant women are more likely to adhere to prophylactic recommendations and actively participate in shared decision-making. This, in turn, enhances patient autonomy and reinforces informed consent practices in antenatal care.
Furthermore, there are notable differences in RhIg dosage regimens and timing across countries, influenced by variations in clinical guidelines, healthcare infrastructure, and product availability. For instance, Norwegian guidelines recommend administering 300 μg (1500 IU), a single dose at 28 weeks of gestation, while in Australia, they employ two-dose regimens (625 IU) at 28 and 34 weeks [11,25,26]. Such inconsistencies can contribute to variability in compliance rates and overall prophylactic effectiveness [20]. Hence, NIPT-guided management improves RhIg compliance by clearly identifying women who require prophylaxis, thereby increasing motivation to complete the recommended dosage schedule and reducing unnecessary administration.
Addressing NIPT and RhIg-associated costs in healthcare
RhD sensitisation remains a major issue, particularly among low socio-economic populations and in developing regions where access to immunoprophylaxis is limited [27]. Therefore, effective cost management is essential in optimising patient care. The findings of our study indicated that, although the incidence of RhD alloimmunisation was low when employing a targeted prophylaxis approach, this strategy was associated with a higher mean cost of patient care compared to routine prophylaxis, with a statistically significant difference (95% CI: 0.11-0.80). High heterogeneity was observed in this study, with I2=100% indicating variability in cost-associated results primarily due to differences in healthcare regulations, prophylaxis guidelines, and RhIg dosing schedules, which are expected given variations in national and regional recommendations. The increased cost observed in the targeted approach can primarily be attributed to the additional expense of foetal RHD genotyping required to determine the foetal RhD status. Unlike routine prophylaxis, which administers RhIg universally to all pregnant women who are RhD-negative, the targeted strategy involves a molecular diagnostic procedure that requires specialised laboratory testing, trained personnel, and advanced equipment. In addition, while the targeted strategy reduces unnecessary RhIg use, the savings from fewer injections often do not outweigh the high upfront cost of genetic testing, especially in settings with limited laboratory infrastructure or low testing volumes [27-30]. Therefore, from a cost perspective, the routine prophylaxis approach remains more economical in many healthcare systems.
Although this approach may initially appear less cost-effective, it offers long-term economic and clinical benefits that support its implementation. NIPT assists in reducing the likelihood of RhD alloimmunisation by precisely identifying RhD-negative foetuses, ensuring that RhD-negative women who carry positive RhD foetuses receive timely and appropriate prophylactic treatment. This precision-based strategy also preserves limited supplies of RhIg, a plasma-derived biological product, for individuals who genuinely require it, thereby promoting more efficient use of healthcare resources [31]. Moreover, by avoiding unnecessary administration of RhIg to women carrying RhD-negative foetuses, the approach minimises exposure to blood products and associated risks, enhances maternal safety and satisfaction, and supports informed decision-making through personalised care [6,28,31]. Further, the gradual reduction in testing costs, improvements in assay efficiency, and economies of scale are expected to make NIPT increasingly affordable in the near future. Studies by shown that as implementation expands, per-test costs decline significantly, ultimately offsetting initial expenditure. When combined with digital health record integration and automated reporting, the workflow efficiency of NIPT-based programs can improve, enhancing cost-effectiveness across diverse healthcare systems [28].
Future economic evaluations should also incorporate indirect cost benefits, such as reduced hospital admissions for HDFN management, decreased neonatal intensive care requirements, and improved maternal quality of life. These broader healthcare savings may further justify the adoption of NIPT-based targeted prophylaxis as a sustainable long-term strategy [28]. Over time, as the cost of genetic testing continues to decline and laboratory efficiency improves, the cost-effectiveness and sustainability of NIPT-based targeted prophylaxis are expected to increase, making it a valuable advancement in modern obstetric practice [30].
Sustainability of employing NIPT in maternal care
The use of NIPT for foetal RHD determination can significantly improve the sustainability of immunoprophylaxis programmes. By accurately identifying RhD-negative foetuses early in pregnancy, NIPT assists targeted administration of anti-D prophylaxis solely to mothers in need. This method helps conserve limited donor-derived RhIg supplies, as the global shortage and donor dependency remain ongoing challenges [2]. It also reduces wastage of biological resources and ensures the long-term availability of RhIg, especially for individuals at genuine risk of alloimmunisation [28,31].
In addition, by reducing the number of individuals exposed to donor-derived products, NIPT decreases the potential risk of transfusion-transmissible infections and addresses ethical concerns related to the use of human-derived materials [28]. This approach also promotes responsible healthcare resource utilisation and aids in precision medicine and patient-centred care. Notably, our study showed a lower incidence of RhD alloimmunisation (0.2%) following the implementation of NIPT-guided prophylaxis. This finding demonstrates the positive effect of the NIPT approach on improving clinical outcomes for RhD-negative pregnant women while also supporting the sustainability of the wider immunoprophylaxis programme. Overall, integrating NIPT into standard antenatal care advances personalised medicine and patient-centred approaches, where interventions are tailored to individual risk profiles, thereby enhancing both effectiveness and ethical standards in foetal-maternal health [8,28].
Limitations
When analysing the meta-analysis's results, several limitations need to be considered. The primary limitations were the scarcity of available studies, resulting in less data variation, and the insufficient number of prospective studies. The majority of studies involved were retrospective or model-based and employed a non-randomised approach, which may introduce confounding factors or bias, thereby compromising the internal validity of the results. Several studies used a historical control group, which may introduce bias, as changes in clinical practice, patient care management, and the availability of patient records could influence study results. Therefore, it was presumed that the documentation had been completed correctly previously. Additionally, the sample sizes between intervention and control groups varied considerably across studies, as some used a nationwide population, potentially affecting the reliability and comparability of pooled results. Finally, the economic analyses were mainly model-based, relying on assumptions about pricing and resource utilisation, and may not fully reflect real-world cost fluctuations or differences in healthcare systems.
Conclusion
The results of this systematic review and meta-analysis evidently demonstrate the effectiveness of NIPT in determining foetal RhD status in RhD-negative pregnant women. NIPT-guided management effectively decreases unnecessary RhIg administration and reduces RhD alloimmunisation by accurately identifying women with RhD-positive foetuses. Early determination of foetal RhD status supports personalised maternal care, identifies individuals at risk of sensitisation, and prevents unnecessary RhIg administration. From an economic perspective, the universal RhIg prophylaxis approach remains more cost-effective under current healthcare practices. However, as genotyping technologies become more widely available and cost-efficient, the sustainability and clinical value of NIPT are expected to improve substantially. The evidence presented in this review supports the safe and effective application of NIPT in clinical practice, thereby minimising the incidence of RhD alloimmunisation and optimising the use of RhIg prophylaxis. Further studies are needed to evaluate the long-term clinical effects, cost-efficiency, and feasibility of implementing NIPT across different healthcare settings. Strengthening evidence in these domains will be crucial for developing guidelines, optimising resource allocation, and promoting the wider use of personalised RhD prophylaxis in routine antenatal care.
Acknowledgement
PCP, CS, and HM conducted the systematic review and meta-analysis and contributed equally to the report writing. CS performed the database searches and created figures. HM carried out the risk of bias assessment and prepared the tables. PCP developed the review manuscript and performed data analysis. DEJ supervised the process and edited the final document.
Disclosure/Conflict of Interest
The Authors declare no conflicts of interest.
Funding
The author(s) received no specific funding for this study.
Data Availability Statement
Data are available upon request.
References
- Mbalibulha Y, Muwanguzi E, Mugyenyi GR, Natukunda B. Occurrence of anti-D alloantibodies among pregnant women in Kasese District, Western Uganda. J Blood Med. 2015:125-129.
- Picchiassi E, Di Renzo GC, Tarquini F, Bini V, Centra M, Pennacchi L, et al. Non-invasive prenatal RHD genotyping using cell-free fetal DNA from maternal plasma: An Italian experience. Transfus Med Hemother. 2015 ;42(1):22-28.
- Akolekar R, Finning K, Kuppusamy R, Daniels G, Nicolaides KH. Fetal RHD genotyping in maternal plasma at 11–13 weeks of gestation. Fetal Diagn Ther. 2011;29(4):301-306.
- Akurut C, Silan F, Yalcintepe S, Ozdemir O. The diagnostic accuracy of non-invasive fetal RhD genotyping by using cell-free fetal DNA in maternal plasma. Fam Pract Palliat Care. 2019;4(1):1-6.
- Klein KB. Non-invasive Fetal RhD Type Prediction by Direct qPCR.
- Blanco S, Giacomi VS, Slobodianiuk LG, Frutos MC, Carrizo LH, Fanin GE, et al. Usefulness of non-invasive fetal RHD genotyping towards immunoprophylaxis optimization. Transfus Med Hemother. 2018;45(6):423-428.
- Manfroi S, Calisesi C, Fagiani P, Gabriele A, Lodi G, Nucci S, et al. Prenatal non-invasive foetal RHD genotyping: diagnostic accuracy of a test as a guide for appropriate administration of antenatal anti-D immunoprophylaxis. Blood Transfus. 2018;16(6):514.
- Londero D, Merluzzi S, Dreossi C, Barillari G. Prenatal screening service for fetal RHD genotyping to guide prophylaxis: the two-year experience of the Friuli Venezia Giulia region in Italy. Blood Transfus. 2022;21(2):93.
- Moise Jr KJ, Gandhi M, Boring NH, O'Shaughnessy R, Simpson LL, Wolfe HM, et al. Circulating cell-free DNA to determine the fetal RHD status in all three trimesters of pregnancy. Obstet Gynecol. 2016;128(6):1340-1346.
- Clausen FB, Rieneck K, Krog GR, Bundgaard BS, Dziegiel MH. Noninvasive antenatal screening for fetal RHD in RhD negative women to guide targeted anti-D prophylaxis. Methods Mol Biol. 2018347-359. New York, NY: Springer New York.
- Sørensen K, Kjeldsen-Kragh J, Husby H, Akkok ÇA. Determination of fetal RHD type in plasma of RhD negative pregnant women. Scand J Clin Lab Invest. 2018;78(5):411-416.
- Macher HC, Noguerol P, Medrano-Campillo P, Garrido-Márquez MR, Rubio-Calvo A, Carmona-González M, et al. Standardization non-invasive fetal RHD and SRY determination into clinical routine using a new multiplex RT-PCR assay for fetal cell-free DNA in pregnant women plasma: results in clinical benefits and cost saving. Clin Chim Acta. 2012;413(3-4):490-494.
- Moussa H, Tsochandaridis M, Jemni-Yacoub S, Hmida S, Khairi H, Gabert J, et al. Fetal RhD genotyping by real time quantitative PCR in maternal plasma of RhD-negative pregnant women from the Sahel of Tunisia. Ann Biol Clin (Paris). 2012;70(6):683-688.
- Wang QP, Dong GT, Wang XD, Gu J, Li Z, Sun AY, et al. An investigation of secondary anti-D immunisation among phenotypically RhD-negative individuals in the Chinese population. Blood Transfus. 2014;12(2):238.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372.
- Analytics C. EndNote reference management software. Philadelphia, PA, USA: Clarivate Analytics; 2025.
- Innovation VH. Covidence systematic review software. Melbourne, Australia: Veritas Health Innovation [Internet]. 2025.
- Vandenbrouckel JP, von Elm E, Altman DG, Gotzsche PC, Mulrow CD, Pocock SJ, et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. PLoS Med. 2007;4(10):1628-1655.
- Sterne JA, Hernán MA, Reeves BC, SavoviÄ? J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355.
- Thorup E, Clausen FB, Brodersen T, Dellgren CD, Ekelund C, Haunstrup TM, et al. Evaluation of the clinical effect of a nationwide implementation of targeted routine antenatal antiâ?D prophylaxis in Denmark. Transfusion. 2025;65(1):29-37.
- Manzanares S, Entrala C, Sánchez-Gila M, Fernández-Rosado F, Cobo D, Martinez E, et al. Noninvasive fetal RhD status determination in early pregnancy. Fetal Diagn Ther. 2014;35(1):7-12.
- Gudlaugsson B, Hjartardottir H, Svansdottir G, Gudmundsdottir G, Kjartansson S, Jonsson T, et al. Rhesus D alloimmunization in pregnancy from 1996 to 2015 in Iceland: A nationâ?wide population study prior to routine antenatal antiâ?D prophylaxis. Transfusion. 2020;60(1):175-183.
- Jensen MP, Damkjær MB, Clausen FB, Ali HA, Hare KJ, Dziegiel MH, et al. Targeted Rhesus immunoglobulin for RhDâ?negative women undergoing an induced abortion: A clinical pilot study. Acta Obstet Gynecol Scand. 2019;98(9):1164-1171.
- Soothill PW, Finning K, Latham T, Wrefordâ?Bush T, Ford J, Daniels G. Use of cff DNA to avoid administration of antiâ?D to pregnant women when the fetus is RhDâ?negative: implementation in the NHS. BJOG. 2015;122(12):1682-1686.
- MacKenzie IZ, Dutton S, Roseman F. Evidence to support the single-dose over the two-dose protocol for routine antenatal anti-D Rhesus prophylaxis: a prospective observational study. Eur J Obstet Gynecol Reprod Biol. 2011;158(1):42-46.
- White SW, Cheng JC, Penovaâ?Veselinovic B, Wang C, White M, Ingleby B, et al. Single dose v twoâ?dose antenatal antiâ?D prophylaxis: a randomised controlled trial. Med J Aust. 2019;211(6):261-265.
- Demirel E, Kelekci S, Ekmekci E, Å?engül M, Iri R, Atasever M. Is the management of Rh-Rh incompatibility with noninvasive fetal Rh genotypingfor targeted prophylaxis cost-effective in the Turkish population?. Turk J Med Sci. 2018;48(1):1-4.
- Darlington M, Carbonne B, Mailloux A, Brossard Y, Levy-Mozziconacci A, Cortey A, et al. Effectiveness and costs of non-invasive foetal RHD genotyping in rhesus-D negative mothers: a French multicentric two-arm study of 850 women. BMC Pregnancy Childbirth. 2018;18(1):496.
- Neovius M, Tiblad E, Westgren M, Kublickas M, Neovius K, Wikman A. Costâ?effectiveness of first trimester nonâ?invasive fetal RHD screening for targeted antenatal antiâ?D prophylaxis in RhDâ?negative pregnant women: a modelâ?based analysis. BJOG. 2016;123(8):1337-1346.
- Mateus-Nino JF, Wynn J, Wiggins-Smith J, Bryant JB, Citty JK, Citty JK, et al. Clinical performance of cell-free DNA for fetal RhD detection in RhD-negative pregnant individuals in the United States. Obstet Gynecol. 2025;145(4):402-408.
- Teitelbaum L, Metcalfe A, Clarke G, Parboosingh JS, Wilson RD, Johnson JM. Costs and benefits of nonâ?invasive fetal RhD determination. Ultrasound Obstet Gynecol. 2015;45(1):84-88.
- Tiblad E, Taune Wikman A, Ajne G, Blanck A, Jansson Y, Karlsson A, et al. Targeted routine antenatal anti-D prophylaxis in the prevention of RhD immunisation-outcome of a new antenatal screening and prevention program. PLoS One. 2013;8(8):e70984.
Citation: Peiris PC, Smith C, Makel H, Jackson DE (2025). Effectiveness of NIPT on RhD-Negative Pregnant Women on RhIg-Induced Isoimmunisation and Associated Healthcare Costs: A Systematic Review and Meta-Analysis. J Blood Disord Transfus. 16:634.
Copyright: © 2025 Peiris PC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.