Indexed In
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- ResearchBible
- Ulrich's Periodicals Directory
- Access to Global Online Research in Agriculture (AGORA)
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- MIAR
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Research Article - (2015) Volume 6, Issue 3
Clinical and Serological Outcomes in Different Surgical Approaches for Hepatic Human Hydatidosis
Abstract
Background: Cystic echinococcosis or hydatidosis occurs as a result of infection by the larval stages of Ecchinococcus granulosus. Treatment approaches of hydatid cysts include the use of albendazole, surgery and/or medico-surgical procedures.
Objective: The present study aimed to investigate and evaluate partial pericystectomy, PAIR technique and combined PAIR technique followed by deroofing, evacuation of cysts and omentoplasty as three therapeutic approaches for treatment of hepatic hydatid cysts.
Methodology: The study was conducted on 54 patients presenting with hepatic hydatid cysts. Patients were included in 3 groups: group I (14 cases) subjected to partial pericystectomy, group II (23 cases) subjected to PAIR technique and group III (17 cases) subjected to PAIR technique followed by deroofing of the cysts and omentoplasty. Diagnosis of hydatid cysts was based on serological testing by ELISA, abdominal ultra-sound examination and parasitological examination of cysts contents. Morbidity, mortality, hospital stay, recurrence and postoperative complications were evaluated for each surgical procedure.
Results: Postoperative bleeding, infection, recurrence and prolonged hospital stay were reported in group I subjected to pericystectomy. Recurrence and postoperative bleeding were reported in group II with no postoperative infection and less hospital stay. No recurrence or postoperative complications were reported in cases included in group III.
Conclusion: Partial surgical procedure with deroofing, evacuation of cysts and omentoplasty performed in this work is recommended as a safe and effective method for entire elimination of parasite with minimal possibility for intra-peritoneal spillage.
Keywords: Hydatid cyst; Partial pericystectomy; PAIR technique; Deroofing
Introduction
Hydatidosis or cystic ecchinococcosis is one of the world’s major zoonotic infections [1]. Larval stages of E. granulosus develop in internal organs of humans and various intermediate hosts as unilocular fluid-filled cystic lesions [2]. Hydatid cysts consist of an internal germinal layer and an outer laminated layer. The parasite cysts gradually expand and cause a granulomatous host reaction, followed by the development of a fibrous tissue layer [3]. Most primary infections consist of a single cyst and are usually asymptomatic for a long period of time, since cyst growth is usually slow. The cysts are usually localized in the liver (75% of cases), although can affect any other organs. The signs and symptoms of liver hydatidosis include hepatomegaly, right/epigastric pain, nausea and vomiting [4].
Abdominal ultrasonography is considered the gold standard for defining the number, site, dimensions and vitality of cysts in addition to its importance in evaluation of treatment options [5]. A definite diagnosis of hydatid cyst is confirmed by identification of precise features as protoscolices, refractile hooks or fragments of a laminated membrane after surgery [6].
There are three therapeutic modalities for the treatment of hepatic hydatid cysts: chemotherapy, PAIR technique and surgery (either open traditional or laparoscopic methods) [7]. Radical cystectomy and hepatectomy approach was recorded to be the traditional mode of treatment, however, in addition to its related prolonged hospitalization; it has a morbidity rate of 14-60%, an overall mortality of up to 7.8% [8]. Operative procedures varying from complete resection to minimal invasive techniques have been developed, but the ideal treatment is still a matter of controversy [9-11]. Several factors affect the preference of therapeutic surgical approach including number and localization of the cysts, surgeon expertise and presence of complications. One of the recommended therapeutic procedures for hepatic hydatid cysts is needle aspiration and concurrent chemotherapy known as the PAIR procedure to be done instead of surgical removal [9,12]. The PAIR procedure is recorded to satisfy all the goals of surgery for hydatid disease by sclerosing germinal membrane and separating laminated membrane with scolicides [13]. Well planned surgical techniques and appropriate procedures based on correct evaluation of disease complications and performance by an experienced team with adequate postoperative support constitute the mainstay of therapy [14].
The present study aimed to investigate clinical and serological outcomes of three lines of treatment of hepatic hydatid cysts. Such lines included surgical removal of the cyst (pericystectomy), PAIR technique and combined PAIR technique followed by de-roofing and evacuation of cysts in addition to omentoplasty.
Patients and Methods
The present study is a cohort conducted on 54 patients presenting with isolated hepatic hydatid cysts and exposed to three different therapeutic interventions and follow up period up to 7 years. They were admitted to the Department of Surgery, National Hepatology and Tropical Medicine Research institute between 2007 and 2014. The cases were divided into three groups according to the performed therapeutic approach as follow I) Retrospective cohort included 14 patients subjected to pericystectomy (data were collected from medical records). II) Prospective cohort included 23 patients exposed to PAIR technique. The third group included 17 cases in a prospective cohort exposed to combined PAIR technique followed by de-roofing and evacuation of cyst contents.
After detailed history and clinical examination, preoperative diagnosis was based on sonography and triphasic CT, serological results in positive cases and postoperative parasitological examination of cysts contents. Cysts' conditions (cyst size, site, number, contents, wall and the surrounding liver parenchyma) and other organs involvement were recorded during abdominal ultrasonographic examination. Patients with hydatid cysts in other organs were excluded from the study, however, management of these cases was done in corresponding departments according to the institutional protocol.
The following data were reported for each admission: age, gender, clinical, imaging evaluation, number, size, and location of cysts, previous surgery for liver hydatidosis, duration of stay, postoperative morbidity, postoperative mortality and recurrence rates. Pre and postoperative investigations including complete blood count, blood group typing and liver function tests were done.
All patients received albendazole tablets in a dose of 400 mg twice daily for two weeks to one month before the therapeutic procedure and for 6 months after therapy.
Surgical Procedures
Pericystectomy
Retrospective analysis of medical records for the first group (14 patients) subjected to radical surgical removal of hepatic hydatid cysts (pericystectomy) was performed. In this intervention, dissection of the cyst along its boundary with healthy liver tissue was done. Blood vessels and small biliary structures passing through the plane between normal liver tissue and the cyst were clamped and divided. Patients' data were recorded to be analyzed.
PAIR technique
In the present study, PAIR technique was done for 23 patients guided by ultrasound visualization under local anesthesia. The PAIR procedure was performed by puncture, aspiration of cyst content, injection of hypertonic saline solution ± 100% alcohol, and re-aspiration of all fluid (PAIR) for small cysts (less than 5 cm) or by a catheterization technique for large sized cysts (larger than 5 cm). Initial puncture was performed by the free hand technique using an 18G needle, through transhepatic approach. The entire fluid in the cyst was aspirated leaving behind only just little fluid to allow visibility of the needle tip. The aspirated 000000000000000was tested by dip stick test (Multistix SG; BAYER Diagnostics) for the presence of bile salts and pigments to rule out biliary communication [15].
Combined surgical procedure
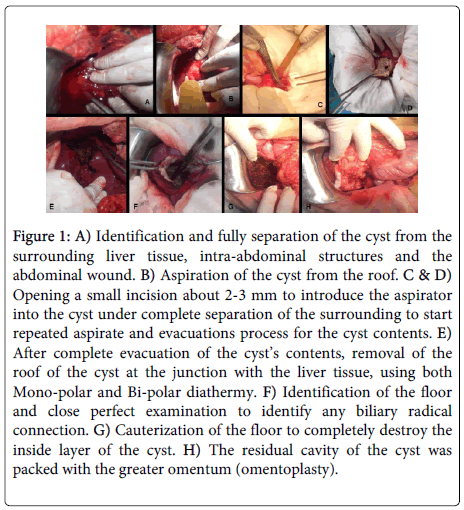
PAIR technique was performed followed by de-roofing and evacuation of the cysts (for the large cysts, more than 5 cm in diameter or failed PAIR technique procedure). The procedure as summarized in figure 1 was done after modification [16]. Under general anesthesia in a supine position for the patient, the abdomen was incised right sub-costal incision either simple or extended to double subcostal or upper midline. A routine intra-operative U/S liver assessment was done. The hepatic cystic lesion was identified and isolated from the surrounding (both from abdominal wall incision, and the intra-abdominal contents viscera using wet towels with hypertonic saline 16 to 20% (isolation was done gradual, first by dry towels then wet with normal saline then wet with 3% hypertonic saline then the last layer with the hypertonic saline 16 to 20%). Affected segment was isolated as previously mentioned. Aspiration of the cyst from the roof by a 10 ml syringe for parasitological and cytology examination was done (Figure 1B). Small incision about 2-3 mm was done to introduce the aspirator into the cyst under complete separation of the surrounding, to aspirate the cyst contents (Figure 1C). After complete aspiration of the cyst content, hypertonic saline 16-20% was injected. The opening was closed and the hypertonic saline was left for 5 to 7 minutes, and then aspirated. The process was repeated then after complete evacuation of the cyst contents, the roof of the cyst was removed at the junction with the liver tissue, using both Mono-polar and Bi-polar diathermy and the Harmonic scalpel. The internal wall of the cyst was explored to detect any biliary radical connection (Figure 1F). Minor biliary radical connections were ligated using Proline 3/0 or 4/0 according to the size of the duct while connections with major bile ducts were managed by intra-operative cholangiography. The internal wall of the cyst was then swabbed with a hypertonic saline and Betadine and then cauterized (Figure 1G) using the mono-polar diathermy (in the coagulation mode with spray mode as well) to destroy completely the floor and the inside layer of the cyst. After complete destruction of the entire of the cyst and good homeostasis, the residual cavity of the cyst was packed with part of the omentum and the cyst was closed with a nearby drain (Figure 1H).
Figure 1: A) Identification and fully separation of the cyst from the surrounding liver tissue, intra-abdominal structures and the abdominal wound. B) Aspiration of the cyst from the roof. C & D) Opening a small incision about 2-3 mm to introduce the aspirator into the cyst under complete separation of the surrounding to start repeated aspirate and evacuations process for the cyst contents. E) After complete evacuation of the cyst’s contents, removal of the roof of the cyst at the junction with the liver tissue, using both Mono-polar and Bi-polar diathermy. F) Identification of the floor and close perfect examination to identify any biliary radical connection. G) Cauterization of the floor to completely destroy the inside layer of the cyst. H) The residual cavity of the cyst was packed with the greater omentum (omentoplasty).
Parasitological examination
Direct parasitological examination: hepatic cysts removed by surgical procedures were examined macroscopically for size, shape, then dissected and the layers were examined properly for the characteristic shape of the germinal layer.
Aspirated fluids from operated or aspirated cysts were examined microscopically as wet unstained mounts (multiple smears were examined for each sample) and resuspended pellets after centrifugation at 500 xg for 10 minutes to detect distinctive scolices or hooks.
Serological investigation: Serum samples were obtained from patients included in the present study before therapeutic intervention and during follow up visits. Samples were tested for anti-hydatid antibodies using ELISA anti-Echinococcus IgG Human ELISA Kit, Abcam, USA.
Follow up of patients
Group I was retrospectively followed through their medical records. Initially the cohorts in prospective works were followed up twice a month for 3 months, followed by once a month for 1 year and then every 3-6 months for the rest of the follow up period. The residual cavity, possible infection and any recurrence were investigated during the follow up visits. Hence, patients were subjected to detailed history taking, clinical examination, ultrasonography, Triphasic- CT abdomen and serological tests.
Ethical considerations: Informed written consent was individually signed by each patient before inclusion in the present study. The current study was conducted according to the institutional ethical and professional guide lines in management and follow up of the cases.
Statistical analysis
All data were statistically analyzed using SPSS version 16 for Windows (SPSS, Chicago,Ill., USA). P values ≤ 0.05 were considered statistically significant.
Results
A total of 54 patients with hepatic hydatid cysts were managed by different therapeutic approaches and included in the current study between 2007 and 2014. There were 19 females and 35 males aged between 22 and 54 years (median 34 years). The significant (p<0.05) predominant clinical presentation was the abdominal pain in 45 patients (83.3%). Other, however, insignificant manifestations as allergic manifestations were pronounced in 9 patients (16.7%) in addition to accidentally discovered cysts in 6 (11.1%) patients during routine investigations. Complete blood count was done for all patients included in the present study and eosinophilia, defined as more than 500 eosinophils per cubic millimeter, was found in17 out of the 54 patients (31.5%) (Table 1). Liver functions were normal in all included cases and ELISA test was positive in 39 patients (72.2%) before intervention. After therapeutic intervention, 5 cases (35.7%) in group I showed disturbed liver enzymes while no changes in the liver enzymes were recorded in other groups (Table 2).
| Main clinical manifestations and investigations | Number of cases | Percentage |
|---|---|---|
| Abdominal pain | 45 | 83.3% |
| Allergic manifestations | 9 | 16.7% |
| Accidental discovery | 6 | 11.1% |
| Eosinophilia | 17 | 31.5% |
| +ve antibodies by ELISA | 39 | 72.2% |
Table 1: Clinical manifestations and investigations done to all cases prior to surgical intervention (original table).
| Group I N=14 cases | Group II N=23 | Group III N=17 | |
|---|---|---|---|
| Preoperative data | |||
| +ve antibodies by ELISA | 10 (71.4%) | 16 (69. 6%) | 13 (76.5%) |
| Disturbed liver enzymes | None | None | None |
| Postoperative data | |||
| +ve antibodies by ELISA (3 months after surgery) | 14 (100%) | 23 (100%) | 17 (100%) |
| Disturbed liver enzymes | 5(35.7%) | None | None |
| Hospital stay time | 10-21 days | 1-2 days | 2-4 days |
| Postoperative bleeding | 2 (14.3%) | None | None |
| Postoperative wound infection | 4 (28.6%) | 3 (13%) | 1 (5.9%) |
| Drains | 3 cases needed 1 drain | No drains were used | 2 drains were used in 3 cases and one drain in 14 cases |
| Recurrence | 2 (14.3%) | 8 (34. 8%) | None |
| Mortality | 3 (21.4%) | None | None |
Table 2: Pre and postoperative data in the three studied groups (original table). As demonstrated in this table, the hospital stay in patients of group I (radical pericystectomy) ranged from 10-21 days and was statistically higher than the other groups (p value<0.001). Patients subjected to PAIR technique stayed at hospital for 1-2 days while patients who were managed by the modified technique stayed at hospital for 2-4 days. No biliary communication was recorded in all patients.
For patients in group I (14 cases managed by pericystectomy), postoperative bleeding occurred in 2 cases (14.3%) and postoperative infection occurred in 4 cases (28.6%). Concerning recurrence, it occurred in 2 cases (14.3%) in group I while 3 cases (21.4%) died shortly after operation due to anaphylactic shock resulting from ruptured cysts. On the other hand, recurrence occurred in 8 cases (34. 8%) out of 23 cases included in group II subjected to PAIR technique while no recurrence was reported in cases of the third group subjected to the modified partial cystectomy (de-roofing) technique.
In group III patients, two drains were placed in wounds of 3 (17.6%) out of 17 cases patients while in the remaining 14 patients (82.4%), one drain was left. The duration of draining ranged from 2-4 days with an average of 3 days. No serious complications or life threatening conditions were reported in any of these 17 patients. Postoperative wound infection was recorded in one patient (5.8%) in group III, while it occurred in four cases (28.57%) in group I and three cases (13 %) group II.
Parasitological examination revealed spherical cysts ranging from 5.3 x 6.8 to 11.2 x 13.4 centimeters in diameter. The characteristic shape of germinal layer was observed and aspiration of the contents of the cysts revealed colorless, clear and gelatinous fluid. Protoscolices of E. granulosus were observed on microscopic examination of the aspirated cysts content.
Discussion
Liver is considered the most frequently affected organ (50-70%) by cystic echinococcosis [11]. Rupture of hydatid cysts can lead to anaphylaxis or dissemination [17,18]. Secondary infection and pressure to nearby organs after reaching remarkable sizes were observed as well. Avoidance of probable complications can be achieved by treatment of hydatid disease even in asymptomatic cases [19].
In general, diagnosis of the hydatid infection requires a variety of laboratory investigations and imaging techniques to ascertain the clinical suspicion. The results of serological examinations for specific antibodies should constantly be linked to different diagnostic parameters and negative serological tests do not exclude infection with hydatid cyst [20]. This agrees with our work, as only 39 cases (72.2%) out of the 54 cases included in the study had positive ELISA readings. The immunological diagnostic methods show variable results, therefore proving insufficient as a single test for the exact diagnosis [21]. In the present study, a definite diagnosis of hydatid cyst was confirmed by direct parasitological examination and identification of protoscolices and refractile hooks.
Therapeutic options for cystic echinococcosis are surgery, chemotherapy, and interventional procedures [22]. However, albendazole has been found ineffective in many cases [23]. A study on the outcome of medical treatment of hydatid cyst in 1000 patients revealed good response in only 30%, decrease in size of the cysts in 30-50% while, no changes reported in 20-40% [22]. However, almost all investigators recommended the use of albendazole prior to any surgical intervention [11]. The present work followed the recommended regimen of albendazole administration, in a dose of 400mg twice daily for at least two weeks before the procedure and for 6 months after surgery [24]. Albendazole has been administered to patients prior to surgery to facilitate safe surgical manipulation as albendazole inactivates protoscolices, changes the integrity of cystic membranes, reduces intra-cystic pressure and reduces the turgidity of the cysts. Therefore, administration of albendazole can minimize the risk of intra-operative spread of scolices and hence reduces risk of possible recurrence [25,26].
On the other hand, several surgical techniques, ranging from hepatic resection to simple cyst evacuation and partial pericystectomy have been used for the treatment of hepatic hydatid cysts. These procedures are eventually targeting the entire eradication of the parasite for complete cure without recurrence. However, the technical procedures are still a matter of controversy [27].
Conservative operations, although easy, have been criticized for a high frequency of postoperative bile leaks and disease recurrence [28]. This agrees with the current work where recurrence was recorded in 8 cohort cases subjected to PAIR technique. PAIR was assumed by some authors to be contraindicated due to anaphylaxis and spillage of scolices, [12] yet other investigators have reported neither anaphylaxis nor dissemination [29].
In the present work, mortality of 21.4% and morbidity up to 28.6% were recorded in group I subjected to radical pericystectomy. Death occurred due to postoperative septic complications and anaphylaxis. Similarly, an overall morbidity of 20 % with total pericystectomy and hepatic resection was recorded [30]. Most of the reported deaths in previous studies resulted from postoperative septic complications and anaphylaxis [31].
No mortality and significantly lower morbidity were reported among cases included in the third group. Mortality and morbidity were remarkably higher with radical procedures than conservative surgical options. Moreover, many surgeons considered these radical methods are too radical for a benign disease [11]. The meta-analysis done by other investigators revealed a mortality of 2%, morbidity of 23% and recurrence rate of 10.4% for such radical types of operations [27]. These authors considered the procedures that remove the parasite and keep the pericyst are easier to carry out than radical ones.
Recurrence is one of the most significant problems in hydatid cyst surgery which occurs in 10 to 30% of patients [32-34] and when recurrent disease occurs, further surgery is probably associated with increasing operative morbidity and mortality [35]. Radical procedures have a higher operative risk and a lower risk of recurrence than conservative procedures which have a higher possibility of recurrence. Recurrence occurs due to inadequate cyst removal or previously undetected cysts [36].
In the current study, no recurrence of cysts was reported during the 7 years follow up period concerning the 17 cases included in group III who experienced partial cystectomy and omentoplasty. In this procedure, different precautions were considered to confirm diagnosis, avoid leakage and to sterilize the cysts’ contents before opening the cystic lesion. In addition to preoperative medical treatment, PAIR technique and parasitological diagnostic approval, intra- operative aspiration and injection of scolicidal agent under strict isolation of abdominal wall and viscera was done using operative packs soaked with scolicidal agent.
Application of dry operative packs in the present work, followed by gradually increasing the concentration of the scolicidal agent within the packs may explain absence of systemic or local toxicity recorded following use of formalin with previous workers [17]. In addition, local toxicity in the form of adhesions using cetrimide and sclerosing cholangitis was recorded following the use of formalin, ethanol, hypertonic saline and silver nitrate [37]. Partial cystectomy and omentoplasty helped our patients to avoid development of biliary fistula.
In the present study, partial procedure (group III) was superior to radical surgical procedures with less morbidity, mortality and decreased hospital stay. The length of hospital stay was significantly higher in the radical procedure than the other techniques. In general, hospital stay differs in various studies according to the technique e.g. percutaneous approach (1-4 days) [11]. On the other hand, length of hospital stay after surgical treatment differed from 6 to 15 days in numerous studies [15,37,38].
In order to monitor remaining or relapsing hydatid cysts, some investigators believed in the usefulness of using ELISA or other serological techniques as a valuable tool not only in diagnosis, but also in follow-up period after surgical applications [39]. In our work, anti hydatid antibodies assessment by ELISA technique detected positive readings in serum of the cases in the postoperative follow up period limiting its usefulness in postsurgical monitoring. Similarly, different immunoglobulins isotypes were detected 3 years after surgical removal of cysts in patients showing no evidences of remaining cysts [40,41].
In conclusion, hydatid disease remains a significant public health problem, however, neglected in endemic areas including Egypt. A diagnostic approach to hepatic hydatid disease should involve a combination of imaging and laboratory techniques in addition to serological analysis. Chemotherapy alone is not sufficient for the treatment of hydatidosis, but should be combined with the preferred surgical option to reduce the recurrence due to spillage of the cyst content during interventions. The main goal of the treatment should be the elimination of the parasite without recurrence and with minimal morbidity and mortality. Although surgical procedures have been the gold standard therapeutic option for hydatidosis, there is still an ongoing controversy about the optimal way of surgery. Partial surgical procedure performed in the current wok is recommended as a safe, simple, and effective and meets all criteria of surgical treatment for hydatid disease: entire elimination of the parasite without destruction of healthy tissue and with all the precautions that prevent intraoperative spillage.
Conflicts of interests
The authors declare that there is no conflict of interests
References
- Lawn SD, Bligh J, Craig PS, Chiodini PL (2004) Human cystic echinococcosis: evaluation of post-treatment serologic follow-up by IgG subclass antibody detection. Am J Trop Med Hyg 70: 329-335.
- Zhang W, Li J, McManus DP (2003) Concepts in immunology and diagnosis of hydatid disease. Clin Microbiol Rev 16: 18-36.
- Thompson RC, McManus DP (2001) Aetiology: parasites and life cycles In: Eckert J, Gemmell M, Meslin FX, Pawlowski Z. WHOI/OIE manual on echinococcosis in humans and animals: a public health problem of global concern. Paris: World Organisation for Animal Health: 1-19.
- Nunnari G, Pinzone MR, Gruttadauria S, Celesia BM, Madeddu G, et al. (2012) Hepatic echinococcosis: Clinical and therapeutic aspects. World J Gastroenterol 18: 1448-1458.
- Pawlowski Z, Eckert J, Vuitton DA, Ammann RW, Kern P, et al. (2001) Echinococcosis in humans: clinical aspects, diagnosis and treatment. In: Eckert J, Gemmell MA, Meslin FX, Pawlowski Z (editors). WHO/OIE Manual on Echinococcosis in humans and animals. Paris: Office International des Epizooties: 20-71.
- Fraiji EK, Connor DH (1997) Echinococcosis. In: Connor DH, Chandler FW, Schwartz DA, Manz HJ, Lack EE (eds.) Pathology of infectious disease. Stamford: Appleton & Lange: 1405-1413.
- Brunetti E, Kern P, Vuitton DA; Writing Panel for the WHO-IWGE (2010) Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop 114: 1-16.
- Ekrami Y (1976) Surgical treatment of hydatid disease of the liver. Arch Surg 111: 1350-1352.
- Khuroo MS, Wani NA, Javid G, Khan BA, Yattoo GN, et al. (1997) Percutaneous drainage compared with surgery for hepatic hydatid cysts. N Engl J Med 337: 881-887.
- Alfieri S, Doglietto GB, Pacelli F, Costamagna G, Carriero C, et al. (1997) Radical surgery for liver hydatid disease: a study of 89 consecutive patients. Hepatogastroenterology 44: 496-500.
- Akkucuk S, Aydogan A1, Ugur M1, Yetim I1, Davran R2, et al. (2014) Comparison of surgical procedures and percutaneous drainage in the treatment of liver hydatide cysts: a retrospective study in an endemic area. Int J Clin Exp Med 7: 2280-2285.
- Smego RA Jr, Sebanego P (2005) Treatment options for hepatic cystic echinococcosis. Int J Infect Dis 9: 69-76.
- Bastid C, Azar C, Doyer M, Sahel J (1994) Percutaneous treatment of hydatid cysts under sonographic guidance. Dig Dis Sci 39: 1576-1580.
- Prousalidis J, Kosmidis C, Anthimidis G, Fachantidis E, Harlaftis N, et al. (2008) Forty-four years' experience (1963-2006) in the management of primarily infected hydatid cyst of the liver. HPB (Oxford) 10: 18-24.
- Rajesh R, Dalip DS, Anupam J, Jaisiram A (2013) Effectiveness of puncture-aspiration-injection-reaspiration in the treatment of hepatic hydatid cysts. Iran J Radiol 10: 68-73.
- Sayek I, Yalin R, Sanaç Y (1980) Surgical treatment of hydatid disease of the liver. Arch Surg 115: 847-850.
- Kouraklis G, Dosios T, Glinavou A, Kouvaraki M, Karatzas G (2001) An alternative approach for the surgical management of hydatid disease of the liver. Langenbecks Arch Surg 386: 62-64.
- Polat FR (2012) Hydatid cyst: open or laparoscopic approach? A retrospective analysis. Surg LaparoscEndoscPercutan Tech 22: 264-266.
- Giorgio A, Di Sarno A, de Stefano G, Liorre G, Farella N, et al. (2009) Sonog-raphy and clinical outcome of viable hydatid liver cysts treated with double percutaneous aspiration and ethanol injection as first-line therapy: efficacy and long-term follow-up. Am J Roentgenol 193: W186-W192.
- Ciobanca PT, Junie ML (2011) Diagnosis confirmation of human cystic echinococcosis by imagistic methods and immunoserological determinations. Sci Parasitol 12: 151-161.
- Ignatov V, Shterev S, Tonev A, Kolev N, Encheva E, et al. (2014) Diagnostics of liver hydatid disease. Scripta Scientifica Medica 46: 37-40.
- Kapan S, Turhan AN, Kalayci MU, Alis H, Aygun E (2008) Albendazole is not effective for primary treatment of hepatic hydatid cysts. J Gastrointest Surg 12: 867-871.
- Tappe D1, Stich A, Frosch M (2008) Emergence of polycystic neotropical echinococcosis. Emerg Infect Dis 14: 292-297.
- CDC. Echinococcosis Treatment Information". November 29, 2013. Retrieved 20 March 2014."The Medical Letter (Drugs for Parasitic Infections)" (PDF).
- Wen H, New RR, Craig PS (1993) Diagnosis and treatment of human hydatidosis. Br J Clin Pharmacol 35: 565-574.
- Smego RA Jr, Bhatti S, Khaliq AA, Beg MA (2003) Percutaneous aspiration-injection-reaspiration drainage plus albendazole or mebendazole for hepatic cystic echinococcosis: a meta-analysis. Clin Infect Dis 37: 1073-1083.
- Buttenschoen K, CarliButtenschoen D (2003) Echinococcus granulosus infection: the challenge of surgical treatment. Langenbecks Arch Surg 388: 218-230.
- Agarwal S, Sikora SS, Kumar A, Saxena R, Kapoor VK (2005) Bile leaks following surgery for hepatic hydatid disease. Indian J Gastroenterol 24: 55-58.
- Yasawy MI, Mohammed AE, Bassam S, Karawi MA, Shariq S (2011) Percutaneous aspiration and drainage with adjuvant medical therapy for treatment of hepatic hydatid cysts. World J Gastroenterol 17: 646-650.
- Birnbaum DJ, Hardwigsen J, Barbier L, Bouchiba N, Le Treut YP (2012) Is hepatic resection the best treatment for hydatid cyst? J Gastrointest Surg 16: 2086-2093.
- Casado AO, Gonzalez ME, Segurola LC, Calvo GA, Pinto GI, et al. (2001) Results of 22 years of experi¬ence in radical surgical treatment of hepatic hydatid cysts. Hepatogastroenterology 48: 235-243.
- Yagci G, Ustunsoz B, Kaymakcioglu N, Bozlar U, Gorgulu S, et al. (2005) Results of surgical, laparoscopic, and percutaneous treatment for hydatid disease of the liver: 10 Years experience with 355 patients. World J Surg 29: 1670-1679.
- Safioleas MC, Misiakos EP, Kouvaraki M, Stamatakos MK, Manti CP, et al. (2006) Hydatid disease of the liver: a continuing surgical problem. Arch Surg 141: 1101-1108.
- El Malki HO, El Mejdoubi Y, Souadka A, Zakri B, Mohsine R, et al. (2010) Does primary surgical management of liver hydatid cyst influence recurrence? J Gastrointest Surg 14: 1121-1127.
- Symeonidis N, Pavlidis T, Baltatzis M, Ballas K, Psarras K, et al. (2013) Complicated liver echinococcosis: 30 years of experience from an endemic area. Scand J Surg 102: 171-177.
- Ammann RW, Eckert J (1996) Cestodes. Echinococcus. Gastroenterol Clin North Am 25: 655-689.
- Mastrandrea S1, Stegel G, Piseddu T, Ledda S, Masala G (2012) A retrospective study on burden of human echinococcosis based on Hospital Discharge Records from 2001 to 2009 in Sardinia, Italy. Acta Trop 123: 184-189.
- Rozanes I, Guven K, Acunas B, Emre A (2007) Cystic echinococcal liver disease: new insights into an old disease and an algorithm for therapy planning. Cardiovasc Intervent Radiol 30: 1112-1116.
- Dessaint JP, Bout D, Wattre P, Capron A (1975) Quantitative determination of specific IgE antibodies to Echinococcus granulosus and IgE levels in sera from patients with hydatid disease. Immunology 29: 813-823.
- Bulut V, Ilhan F, Yucel AY, Onal S, Ilhan Y, et al. (2001) Immunological follow-up of hydatid cyst cases. MemInstOswaldo Cruz 96: 669-671.
- Doiz O, Benito R, Gil J, Rojas A, Rubio MC, et al. (2002) Pre- and postsurgical detection of IgG, IgM, and IgA specific to hydatidosis by ELISA with purified antigen enriched with the 5/B antigen complex. J Clin Lab Anal 16: 295-298.
Copyright: © 2015 Abdelraouf, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.